NF-κB: Unraveling the Master Regulator of Immune and Inflammatory Responses
Conclusion: A Masterpiece of Complexity
The intricate symphony of the human immune system is a marvel of biological engineering, orchestrating a complex defense against pathogens while maintaining a delicate balance to prevent self-attack. At the heart of this system lies a master regulator, a transcription factor family that dictates responses ranging from immediate defense to long-term immune memory. Its name, Nuclear Factor kappa-light-chain-enhancer of activated B cells, hints at its discovery within immune cells, but its influence extends far beyond. It acts as a central hub, controlling the expression of a vast number of genes critical for immunity, inflammation, cell survival, and proliferation. Understanding it is therefore paramount to comprehending cellular defense mechanisms and the pathogenesis of numerous diseases.
Understanding the Role of NF-κB in Immune Response
It represents a family of transcription factors that play a pivotal role in cellular responses. Initially identified for its function in B lymphocytes, where it binds to the enhancer element of the kappa light chain gene, its broader significance quickly became apparent. It is a potent regulator of gene expression, influencing a wide array of cellular processes fundamental to survival, growth, and immunity. Its activation is rapid and tightly controlled, allowing cells to respond swiftly to a diverse range of stimuli, including infections, stress, and inflammatory signals.
The family in mammals comprises five distinct proteins: RelA (also known as p65), RelB, c-Rel, NF-κB1 (which generates p50 and its precursor p105), and NF-κB2 (generating p52 and its precursor p100). These subunits share a highly conserved domain known as the Rel Homology Domain (RHD). This domain is crucial for their ability to dimerize with one another and for their direct binding to specific DNA sequences, known as κB sites, located in the promoter or enhancer regions of target genes. The interplay between these subunits and their dimerization partners determines the specific transcriptional outcomes and cellular functions that they regulate.
It serves as a critical nexus in the intricate network of cellular signaling. Its activation is a hallmark of inflammatory processes, mediating the production of crucial signaling molecules called cytokines, chemokines, and adhesion molecules that recruit immune cells to sites of injury or infection. Beyond inflammation, it is fundamental to the development, function, and survival of immune cells, including lymphocytes. It also plays a significant role in non-immune processes such as cell proliferation, differentiation, and apoptosis. The dysregulation of this signaling is implicated in a vast number of diseases, from chronic inflammatory conditions and autoimmune disorders to cancer and metabolic diseases, underscoring its central importance in health and disease.
This article will delve into the multifaceted world of NF-κB, dissecting its core components, activation pathways, and regulatory mechanisms. We will explore the distinct roles of its various subunits, followed by a detailed examination of the canonical and non-canonical signaling pathways. The sophisticated layers of regulation, particularly through post-translational modifications, will be illuminated. Furthermore, we will touch upon the emerging understanding of its presence within mitochondria and its profound impact on cellular metabolism. Finally, we will discuss the critical balance it strikes in health and disease, and the therapeutic strategies being developed to modulate its activity.
The family’s diverse subunits provide a flexible platform for cellular regulation. Each subunit can form homodimers or heterodimers, and these combinations exhibit distinct DNA-binding affinities and transcriptional activities. This molecular plasticity allows it to fine-tune cellular responses based on the specific signal received and the cellular context.
The five core subunits—RelA, RelB, c-Rel, NF-κB1, and NF-κB2—are the building blocks of dimers. RelA (p65) and c-Rel are considered “transactivation-capable” subunits due to the presence of a potent transactivation domain, while NF-κB1 (p50) and NF-κB2 (p52) are generally considered “transactivation-null” as they lack a strong transactivation domain and can even possess repressive functions. RelB is another transactivation-capable subunit. The precursor forms, p105 and p100, are processed through ubiquitination and proteolysis to yield the mature p50 and p52 subunits, respectively. This processing itself is a regulatory step that influences the final repertoire of dimers.
The Rel Homology Domain (RHD) is the unifying structural feature across all subunits. This domain is responsible for two critical functions: dimerization, allowing subunits to pair up to form homodimers or heterodimers, and direct binding to specific DNA sequences (κB sites). The RHD’s ability to mediate these interactions is the fundamental basis for its role as a transcription factor. The specific composition of the dimer dictates which κB sites it can bind to and what transcriptional outcome it elicits.
The most extensively studied dimer is the heterodimer composed of p50 (from NF-κB1) and RelA (p65). This complex is highly abundant and plays a central role in orchestrating inflammatory responses. However, numerous other heterodimers exist, such as p50/c-Rel, p52/RelB, and p50/RelB. Each combination possesses unique properties that allow it to engage in a diverse number of cellular functions. For instance, dimers containing p50 can often bind DNA with high affinity, while the presence of RelA or c-Rel recruits co-activators that enhance gene transcription.
The individual subunits contribute distinct functional nuances. RelA (p65) is crucial for driving the expression of a wide array of inflammatory genes due to its strong transactivation domain. c-Rel is also a potent activator but has been implicated in distinct roles, including cell survival and immune cell development, and its expression pattern can differ from RelA. The p50 subunit, while often acting as a partner to RelA, can also form homodimers that primarily repress gene transcription or have more specialized roles. RelB, typically dimerizing with p52, is particularly important in the development and function of lymphoid tissues and dendritic cells.
The canonical, or classical, pathway is the primary route for rapid inflammatory responses. It is triggered by a multitude of extracellular stimuli and is characterized by swift activation, typically occurring within minutes. This pathway is essential for mounting an immediate defense against invading pathogens and cellular damage, initiating acute inflammation.
The canonical pathway is activated by various molecular cues. These include pathogen-associated molecular patterns (PAMPs) recognized by pattern recognition receptors (PRRs) like Toll-like receptors (TLRs) and RIG-I, as well as endogenous danger signals such as the pro-inflammatory cytokine IL-1β and other cytokines. These initial signals converge on intracellular signaling complexes that ultimately lead to the activation of the IKK complex.
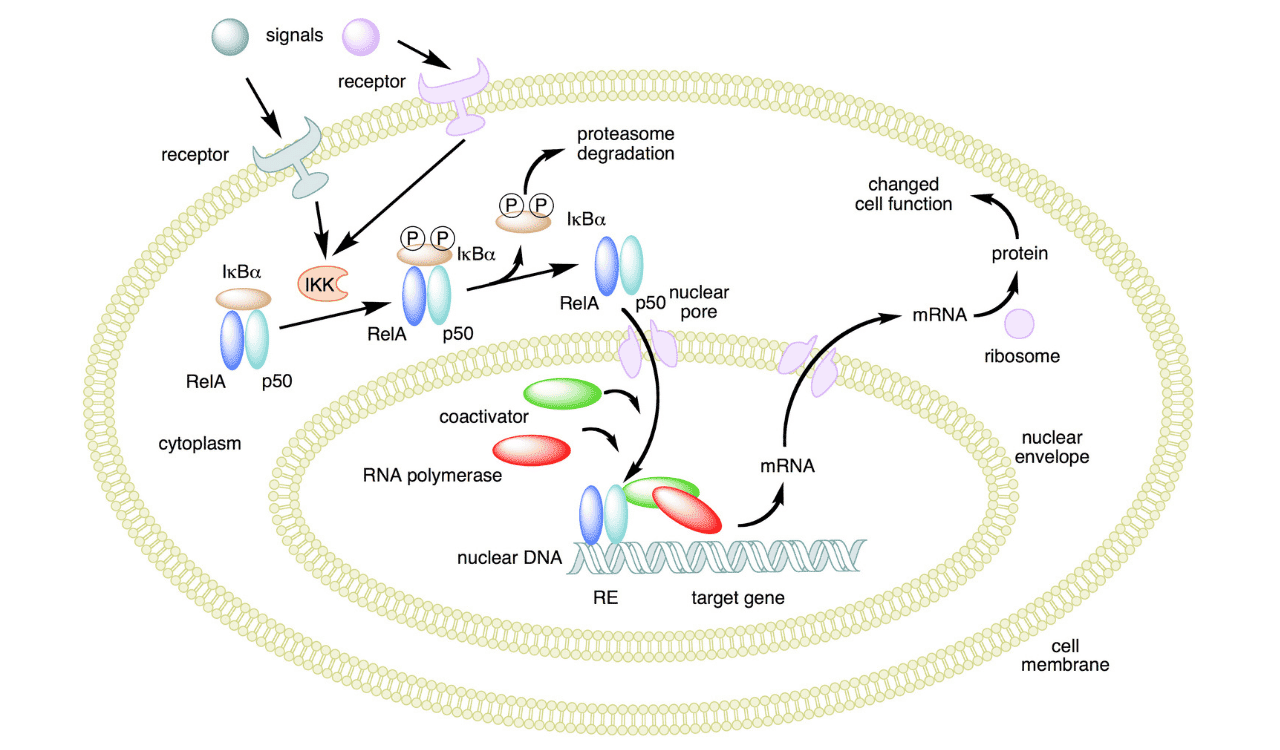
The pivotal event in canonical activation is the formation and activation of the IκB kinase (IKK) complex. This complex typically comprises two catalytic subunits, IKKα and IKKβ, and a regulatory subunit, NEMO (NF-κB essential modulator). While IKKα can homodimerize and mediate specific functions, the IKKβ subunit is largely responsible for phosphorylating the inhibitor of kappa B (IκB) proteins in the canonical pathway. NEMO is critical for transducing upstream signals, particularly those involving ubiquitination, to activate the kinase activity of IKKβ.
In the resting state, dimers are sequestered in the cytoplasm by their inhibitory proteins, primarily IκBα. Upon stimulation, the activated IKK complex phosphorylates IκBα at specific serine residues. This phosphorylation event marks IκBα for rapid polyubiquitination by an E3 ubiquitin ligase complex. The polyubiquitinated IκBα is then recognized and degraded by the 26S proteasome. This degradation releases the dimer from cytoplasmic inhibition. This process is remarkably fast, with the entire cascade from stimulus to IκBα degradation occurring within minutes.
Once released from IκBα, the dimer translocates into the nucleus. Here, it encounters its target DNA sequences, the κB sites, within the regulatory regions of specific genes. Through its RHD, the dimer binds to these sites, often in conjunction with other transcription factors and co-activators or co-repressors. This DNA binding event is the crucial step that initiates the transcription of responsive genes, leading to a cascade of cellular activities.
The genes activated by the dimers orchestrate a wide array of critical cellular functions. These include the production of potent inflammatory mediators such as TNF-α, IL-6, and IL-1β, which amplify the inflammatory response and recruit immune cells. It also promotes cell survival by upregulating anti-apoptotic genes and plays a vital role in the development and function of B cells, influencing antibody production and immune memory. Its actions are central to combating infections but can become detrimental if chronically active.
While the canonical pathway governs rapid inflammatory responses, the non-canonical, or alternative, pathway plays a distinct and crucial role, particularly in adaptive immunity and lymphoid organ development. This pathway is typically activated by a more restricted set of extracellular stimuli and involves a different cascade of events, often leading to slower and more specific cellular outcomes.
The non-canonical pathway is primarily activated by members of the TNF receptor superfamily, most notably RANK (Receptor Activator of Nuclear factor Kappa-B) and CD40. Ligand binding to these receptors triggers intracellular signaling that leads to the activation of a specific IKK complex containing IKKα homodimers. Unlike the canonical pathway that relies on IκBα degradation, the non-canonical pathway involves the processing of precursor subunits.
A key kinase upstream of the IKK complex in the non-canonical pathway is NIK (NF-κB-inducing kinase). Upon receptor activation, NIK is stabilized and becomes active. NIK then phosphorylates and activates the IKKα subunit, leading to its homodimerization and subsequent phosphorylation of the NF-κB2 precursor, p100. This phosphorylation is the critical trigger for the processing of p100.
In the non-canonical pathway, activated IKKα homodimers phosphorylate the precursor protein p100. This phosphorylation event initiates a cascade of ubiquitination and proteolytic processing steps that cleave p100, releasing the active p52 subunit. The p52 subunit then typically dimerizes with RelB, another member of the family.
The newly formed p52/RelB heterodimer, generated through the non-canonical pathway, is then translocated into the nucleus. Similar to the canonical pathway, this nuclear entry allows the dimer to bind to specific κB sites in target genes. However, the genes regulated by the p52/RelB dimer are distinct from those controlled by the canonical pathway, leading to different cellular outcomes.
The non-canonical pathway plays a crucial role in specific aspects of immune development and function. It is essential for the proper formation of secondary lymphoid organs, such as lymph nodes and Peyer’s patches. It also contributes to the development and maturation of T cells and dendritic cells, and it plays a role in maintaining immune homeostasis. Its activation is typically slower and more sustained compared to the canonical pathway, reflecting its involvement in developmental and long-term immune programming.
The regulation of activity extends far beyond the initial activation cascade. A sophisticated array of post-translational modifications (PTMs) acts upon subunits, their inhibitors, and associated signaling proteins. These modifications fine-tune virtually every aspect of function, including its DNA-binding affinity, transcriptional activity, subcellular localization, and protein-protein interactions.
Phosphorylation is a critical PTM that impacts signaling at multiple levels. The IKK complex’s phosphorylation of IκBα is the hallmark of canonical activation. However, subunits themselves, as well as other regulatory proteins, are also targets of various kinases. Phosphorylation can alter the conformation of dimers, influencing their ability to bind DNA, recruit co-activators or co-repressors, and even control their movement between the cytoplasm and the nucleus.
Acetylation, typically occurring on lysine residues, is another key PTM that significantly influences function. Acetylation of subunits, particularly RelA (p65), can enhance their transcriptional activity by promoting interactions with co-activator proteins. It can also affect DNA binding and modulate the protein’s stability and localization. The balance between acetyltransferases and deacetylases is thus crucial for determining the magnitude and duration of mediated gene expression.
Ubiquitination, the attachment of ubiquitin molecules to a protein, is most famously known for its role in targeting IκBα for degradation by the proteasome, thereby enabling canonical activation. However, ubiquitination also plays critical non-proteasomal roles in signaling. For example, certain ubiquitination events can serve as signaling scaffolds, mediating protein-protein interactions essential for signal transduction. Proteins like TRAF6 are heavily involved in ubiquitination events that propagate signals upstream of the IKK complex.
The true complexity of regulation lies in the intricate interplay between these various PTMs. Phosphorylation, acetylation, and ubiquitination do not act in isolation but rather coordinate their actions to precisely control transcriptional output. For instance, a specific phosphorylation event might prime a protein for subsequent acetylation, or ubiquitination might be required for both degradation and signaling. This combinatorial control allows for exquisite fine-tuning of gene expression, ranging from robust activation of inflammatory genes to precise transcriptional silencing when necessary.
It is indispensable for life, acting as a critical guardian of cellular and organismal health. However, its pervasive influence means that its dysregulation is a common denominator in a vast spectrum of diseases. The delicate balance between its protective and detrimental roles is a central theme in understanding human pathology.
In healthy individuals, it is vital for maintaining immune homeostasis. It ensures the appropriate development, survival, and function of immune cells, including T cells and B cells. Its role in promoting cell survival is crucial for preventing the premature loss of these vital immune sentinels. The incidence of global autoimmune diseases has risen yearly by 19.1%, with rheumatological diseases such as Sjogren’s and lupus rising 7.1% per year, highlighting the need for understanding immune dysregulation.
Given its central role in numerous diseases, it represents a highly attractive therapeutic target. A significant number of anti-inflammatory drugs are currently in use and in development, with the global anti-inflammatory therapeutics market size calculated at $109.58 billion in 2025 and predicted to increase to approximately $164.44 billion by 2035, expanding at a CAGR of 4.14% [Nova One Advisor, 2025]. Hospital pharmacies are the largest distribution channel for anti-inflammatory drugs, due to their role in administering high-cost, biologic therapies and managing acute inflammation cases.
It stands as a testament to the exquisite regulatory mechanisms that govern cellular life. From its fundamental role in initiating rapid inflammatory responses via the canonical pathway to its nuanced control over adaptive immunity through the non-canonical pathway, it is an indispensable orchestrator. The intricate tapestry of post-translational modifications further refines its actions, demonstrating a level of biological control that is both powerful and precise. Its pervasive influence, while essential for health and immunity, also positions it at the nexus of numerous diseases, making it a critical target for therapeutic intervention. As research continues to unravel the subtle intricacies of signaling, particularly its emerging roles within mitochondria and its dynamic interplay with cellular metabolism, novel and more targeted therapeutic strategies hold immense promise for tackling a wide range of human ailments, from chronic inflammatory conditions to complex diseases like cancer and neurodegeneration. The ongoing exploration of this master regulator is continually revealing new layers of its complexity and potential for impacting human health.



Leave a Reply