Alpha-Synuclein Seed Amplification Assays: The Game-Changer for Early PD Detection and Clinical Trial Stratification
For decades, the diagnosis of Parkinson’s disease has been a clinical puzzle, pieced together from a patient’s motor symptoms long after the disease has taken root. By the time tremors and rigidity manifest, a significant portion—often over 50%—of the critical dopamine-producing neurons in the brain have already been lost. This late-stage diagnosis has been a formidable barrier to developing treatments that could slow or halt the disease’s progression. The medical and research communities have urgently sought a definitive biological marker—a clear, objective signal of the disease process itself, detectable in its earliest stages. This search has led to a pivotal breakthrough, centering on a protein at the heart of the disease’s pathology, and a technology poised to redefine our entire approach to neurodegenerative disorders.
Introduction: The Urgent Need for Early Parkinson’s Disease Diagnosis
The Elusive Nature of Parkinson’s Disease (PD)
Parkinson Disease has long been defined by its outward signs. A diagnosis traditionally hinges on a neurologist’s evaluation of a patient’s medical history and the presentation of cardinal motor clinical symptoms like bradykinesia (slowness of movement), resting tremor, and rigidity. However, these symptoms are the last domino to fall in a long and destructive chain of neurological events. They only emerge after substantial, irreversible damage has occurred.
This diagnostic lag is compounded by uncertainty. Several other neurodegenerative conditions can mimic the symptoms of early PD, leading to misdiagnosis in up to 20% of cases in early stages, as noted in studies published in journals like Movement Disorders. This ambiguity complicates patient care, delays appropriate management strategies, and critically undermines the integrity of clinical trials for new therapies. The historical absence of definitive diagnostic tests has forced clinicians and patients to navigate a landscape of probability rather than certainty.
The Central Role of Alpha-Synuclein (α-syn) in PD Pathology
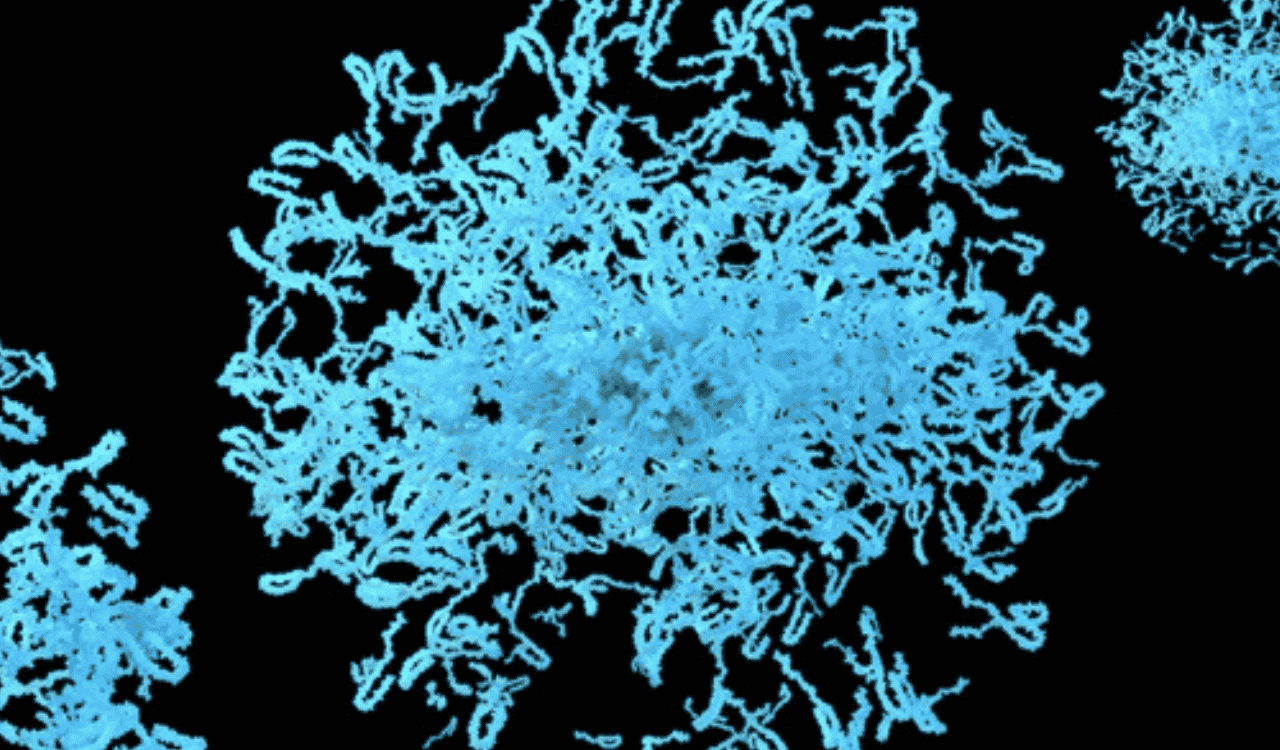
The key to unlocking this diagnostic challenge lies with a protein called alpha-synuclein. In its healthy state, this protein is abundant in the brain and is believed to regulate neurotransmitter release. In Parkinson Disease and a related group of disorders known as synucleinopathies, alpha-synuclein misfolds into a toxic, pathological shape.
This misfolding triggers a catastrophic chain reaction. These small, misfolded aggregates of pathological alpha-synuclein act as “seeds,” corrupting healthy alpha-synuclein proteins and causing them to misfold and clump together. This cascade of αSyn pathology culminates in the formation of large, insoluble inclusions within neurons known as Lewy bodies, the defining pathological hallmark of PD. These toxic protein clumps disrupt cellular function, spread from neuron to neuron, and are believed to drive the relentless neurodegeneration that causes the disease’s devastating symptoms.
Introducing Alpha-Synuclein Seed Amplification Assays (αSyn-SAA): A Paradigm Shift
The discovery of alpha-synuclein’s seeding behavior paved the way for a revolutionary technology: the Alpha-Synuclein Seed Amplification Assay (αSyn-SAA). This groundbreaking laboratory test is not designed to measure the amount of alpha-synuclein, but rather to detect its pathological, seeding-competent form.
By identifying the very “seeds” of the disease, this powerful diagnostic tool can confirm the core pathology of Parkinson’s with unprecedented accuracy, often years before a patient develops motor symptoms. This technology represents a monumental shift from a symptom-based clinical diagnosis to a specific, biological one. As highlighted by an editorial in The Lancet Neurology, this is a “game-changer,” heralding a new era in the fight against neurodegenerative disease.
Unpacking the Science: How αSyn-SAA Works to Detect Pathological Proteins
The elegance of the αSyn-SAA lies in its ability to amplify a nearly imperceptible signal into a robust, measurable one. The technology harnesses the inherent, self-propagating nature of misfolded alpha-synuclein, essentially recreating the disease’s molecular chain reaction in a test tube. Understanding this process requires a look at the “prion-like” behavior of the protein and the clever biochemical techniques developed to exploit it.
The “Prion-Like” Mechanism of Alpha-Synuclein Aggregation
The concept of “prions”—misfolded proteins that can induce normally folded proteins to adopt their pathological shape—was first established in diseases like Creutzfeldt-Jakob disease. Researchers have discovered that alpha-synuclein behaves in a similar “prion-like” manner. A single misfolded α-syn aggregate, or “seed,” introduced into an environment containing healthy α-syn proteins, can act as a template. It corrupts the healthy proteins, causing them to misfold and join the aggregate. This process grows exponentially, creating larger fibrils and spreading the αSyn pathology from cell to cell and throughout the nervous system. It is this specific seeding capacity that is the unique signature of the disease process.
The Principle of Seed Amplification Assays
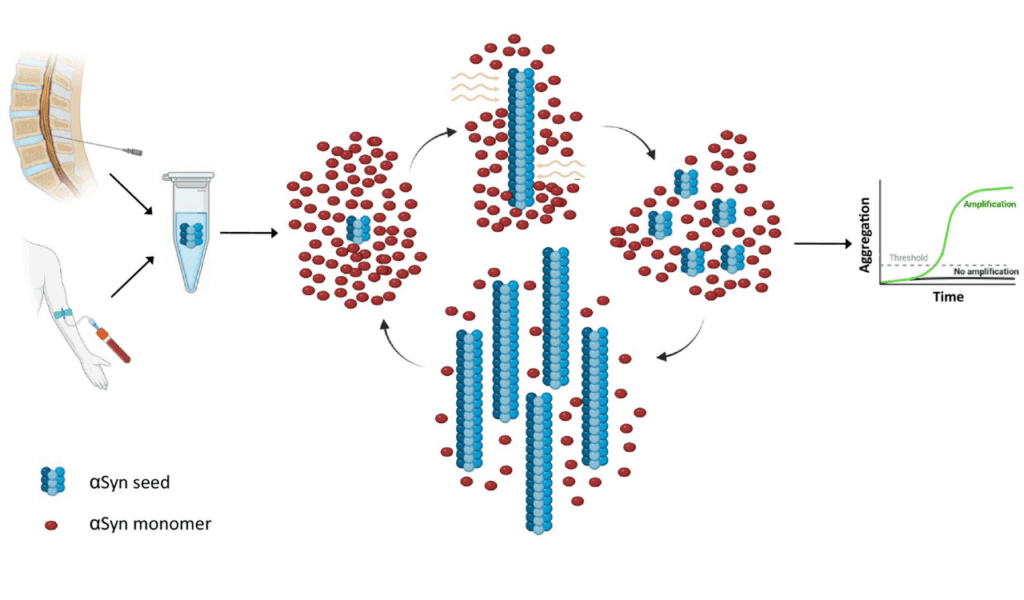
The Seed Amplification Assay is ingeniously designed to exploit this seeding mechanism. The core principle is straightforward: a patient’s biological sample, which may contain infinitesimally small quantities of pathological alpha-synuclein seeds, is added to a reaction mixture containing a large supply of normal, healthy α-syn protein (the substrate).
If pathological seeds are present, they begin converting the healthy substrate into misfolded aggregates. This process is dramatically accelerated through cycles of vigorous shaking (agitation) and incubation. The shaking breaks the newly formed larger aggregates into smaller pieces, each of which can now act as a new seed, exponentially amplifying the reaction. This biochemical chain reaction makes it possible to detect even a single pathological seed from a patient sample.
Key αSyn-SAA Techniques: Real-Time Quaking-Induced Conversion (RT-QuIC)
One of the most prominent and well-validated SAA techniques is Real-Time Quaking-Induced Conversion, or RT-QuIC. In this assay, the reaction is performed in a multi-well plate, and the mixture includes a special fluorescent dye (like Thioflavin T) that binds specifically to the aggregated form of alpha-synuclein.
As the pathological seeds from the patient sample amplify and form new aggregates, the dye binds to them and emits a fluorescence signal. A specialized reader monitors this signal in real-time. A sample from a person with Parkinson Disease will show a characteristic sigmoidal curve of increasing fluorescence, while a sample from an individual without this pathology will remain at baseline. This provides a clear, objective “yes/no” answer about the presence of the disease’s core pathology.
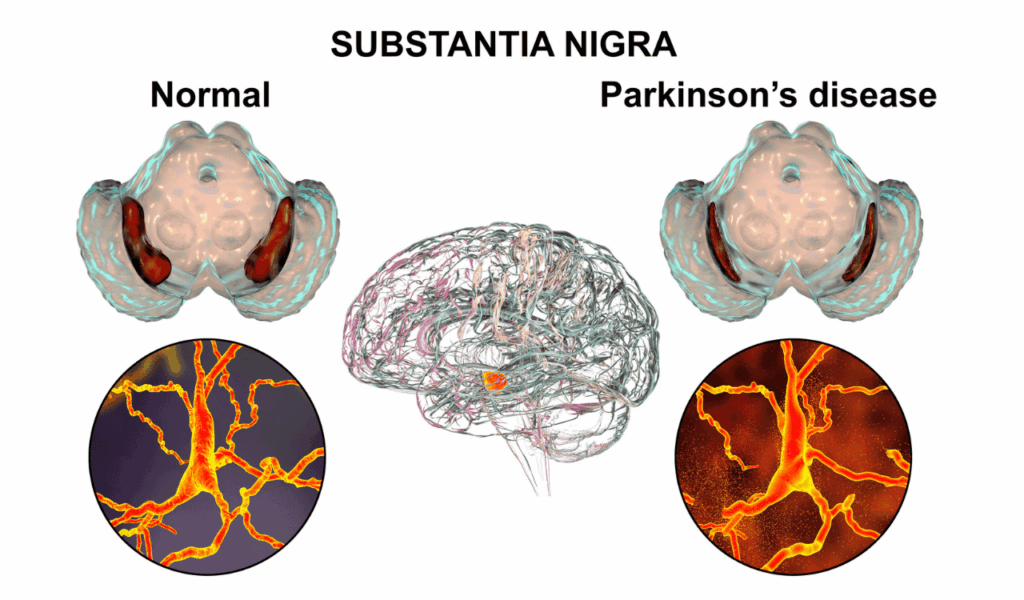
Early and Accurate Diagnosis: Revolutionizing Parkinson’s Detection and Differentiation
The clinical implications of a highly accurate biological test for Parkinson Disease are profound. The αSyn-SAA moves the goalposts for diagnosis from the late, symptomatic stage to the earliest, pre-symptomatic phase of the disease. This capability is not just an incremental improvement; it is a fundamental redefinition of what it means to diagnose a neurodegenerative disorder.
Detecting Parkinson’s Disease in the Prodromal Stage
The “prodromal” stage of Parkinson’s refers to a period that can last for years, or even decades, before classic motor symptoms appear. During this time, patients may experience non-motor symptoms like REM sleep behavior disorder (RBD), constipation, or loss of smell. While these are known risk factors, they are too non-specific to confirm a diagnosis.
However, landmark studies, such as the Parkinson’s Progression Markers Initiative (PPMI), have shown that αSyn-SAA can detect pathological synuclein in at-risk individuals with remarkable accuracy—over 90% in some cohorts. This means the assay can confirm the underlying αSyn pathology long before irreversible motor impairment begins, opening a critical window for potential intervention and achieving an Accurate Diagnosis at the biological level.
Differentiating Parkinson’s from Mimics and Other Synucleinopathies
A significant challenge in neurology is distinguishing true Parkinson Disease from atypical parkinsonian syndromes, such as multiple system atrophy (MSA) or Progressive Supranuclear Palsy (PSP), which have overlapping symptoms but different prognoses and underlying pathologies. αSyn-SAA has demonstrated exceptional specificity, consistently testing negative in patients with non-synuclein-related parkinsonism like PSP.
Furthermore, ongoing research suggests that variations in SAA kinetics—the speed and intensity of the reaction—may even help differentiate between different synucleinopathies like PD, dementia with Lewy bodies (DLB), which is also known as Lewy Body Dementia, and MSA. These diseases are thought to be caused by different “strains” or fibrils conformations of misfolded alpha-synuclein, which may have unique biophysical profiles that the assay can distinguish.
The Profound Impact of an Accurate Diagnosis on Patients and Care
For a patient, receiving a definitive, early diagnosis can be life-changing. It ends the “diagnostic odyssey” of uncertainty, specialist visits, and inconclusive diagnostic tests. An Accurate Diagnosis allows patients and their families to plan for the future, make informed decisions about lifestyle and work, and connect with support networks sooner. Clinically, it ensures the patient receives the most appropriate symptomatic treatments from the outset. Perhaps most importantly, it provides clarity and empowers the patient with knowledge, shifting the dynamic from passive waiting to proactive management.
Transforming Clinical Trial Stratification: A Catalyst for New Therapies
Beyond its diagnostic utility, the αSyn-SAA is poised to have its most significant impact by revolutionizing the design and execution of clinical trials. For years, the development of disease-modifying therapies for Parkinson’s has been hampered by the inability to select the right patients for studies and objectively measure a drug’s effect on the underlying biology.
Precision Patient Stratification for Targeted Clinical Trials
Historically, clinical trials for PD have enrolled participants based on clinical symptoms. This approach has a critical flaw: a cohort of patients with “Parkinson’s symptoms” might include individuals with different underlying pathologies (misdiagnoses) or those at different disease stages. This heterogeneity can mask a drug’s potential benefit. With αSyn-SAA, trial organizers can now definitively confirm that every enrolled participant has the specific alpha-synuclein pathology the drug is designed to target. This precision stratification creates cleaner, more homogeneous patient groups, dramatically increasing the statistical power of a study and its likelihood of success.
Enabling Early Intervention Trials and Disease Modification
The ultimate goal in Parkinson’s research is to find therapies that can slow, stop, or even reverse the disease. To be effective, such treatments must be administered as early as possible. The ability of αSyn-SAA to identify individuals in the prodromal stage is a game-changer for this pursuit. For the first time, researchers can design and conduct clinical trials in pre-symptomatic patient populations who have a confirmed biological diagnosis. This opens the door to testing neuroprotective therapies when they have the greatest chance of preserving brain function and altering the long-term course of the disease.
Objective Monitoring of Therapeutic Efficacy
Another major hurdle in PD drug development is the lack of objective biomarkers to measure whether a treatment is working. Trials have traditionally relied on clinical rating scales, which are subjective and slow to show change. While αSyn-SAA is currently used as a qualitative (positive/negative) test, research is actively exploring its quantitative potential. The hope is that changes in the “seeding activity” measured by the assay over time could serve as a direct, objective biomarker of therapeutic response. For example, a successful drug might lead to a reduction in the SAA signal, providing clear evidence that the treatment is hitting its biological target.
Accessibility and Practical Implementation: Bridging Research to Clinic
For any breakthrough technology to realize its full potential, it must successfully transition from the specialized research laboratory to routine clinical practice. The path for αSyn-SAA involves optimizing sample collection, standardizing procedures, and ensuring broad accessibility.
Current Sample Sources: Cerebrospinal Fluid (CSF) and Associated Considerations
To date, the most reliable and extensively validated sample type for αSyn-SAA is cerebrospinal fluid (CSF). This clear fluid surrounds the brain and spinal cord, providing a direct window into the brain’s biochemical environment. Numerous large-scale studies have demonstrated that αSyn-SAA on CSF can detect Parkinson’s pathology with over 90% sensitivity and specificity.
However, obtaining CSF requires a lumbar puncture, more commonly known as a spinal tap. While this procedure is generally safe when performed by an expert, it is invasive and can be associated with side effects like a post-lumbar puncture headache. This can be a barrier for some patients and has been a primary driver in the search for more accessible sample sources. The processing of Cerebrospinal Fluid Total protein and other components is also a critical step for ensuring assay reliability.
The Promise of Less Invasive Samples
The holy grail for SAA implementation is a reliable test using a less invasive sample. Research is advancing rapidly in this area. Scientists are applying SAA techniques to various tissues where pathological alpha-synuclein can be found, due to its spread to the peripheral nervous system.
Promising results have emerged from samples of the olfactory mucosa (the lining of the nasal cavity), which can be collected via a nasal swab, and from skin biopsies. Other studies are making progress with submandibular gland biopsies and even blood tests, where new methods are being developed to isolate the tiny amounts of α-syn seeds. The validation of a less invasive αSyn-SAA would dramatically lower the barrier to testing, potentially making it a routine part of neurological workups.
The Path Towards Widespread Clinical Integration
Bringing αSyn-SAA into mainstream clinical use requires several key steps. First is standardization. Different labs may use slightly different protocols, leading to variability. Efforts are underway to establish standardized reference materials and quality control procedures. Second is regulatory approval and commercialization, processes that are already in progress. Finally, education for clinicians and patients is crucial to ensure the test is used appropriately and its results are interpreted correctly within the broader clinical context of the patient.
The Future Landscape: Broader Implications and Hope for Neurodegenerative Disorders
The development and validation of αSyn-SAA is more than just a new test for a single disease; it represents a successful proof-of-concept for a new era of biological diagnostics in neurodegeneration. Its impact will ripple across the field, influencing how we diagnose, manage, and research a wide range of related disorders.
Broader Impact Across Synucleinopathies
While Parkinson Disease is the most common synucleinopathy, the family of disorders also includes Dementia with Lewy Bodies (DLB) and Multiple System Atrophy (MSA). These conditions share the common thread of alpha-synuclein pathology but have distinct clinical features. αSyn-SAA is proving to be a powerful tool for diagnosing these disorders as well. As mentioned, subtle differences in assay results may reflect underlying differences in the structure of the α-syn seeds characteristic of each disease, potentially leading to assays that not only detect but also specify the type of synucleinopathy.
Synergizing with Other Biomarkers and Genetic Insights
The future of neurodegenerative care will not rely on a single biomarker but on a comprehensive panel. αSyn-SAA will be a cornerstone, but its power will be magnified when combined with other information. For instance, integrating SAA results with genetic mutations known to increase Parkinson’s risk (such as in the GBA or LRRK2 genes) will provide a richer, multi-dimensional picture of a patient’s disease. Similarly, combining SAA with neuroimaging signatures and other fluid biomarkers (like those for Alzheimer’s) will help disentangle complex cases of mixed dementia, paving the way for truly personalized medicine.
A Horizon of Hope for Patients and Accelerated Research
Ultimately, the most profound impact of αSyn-SAA is the hope it provides. For patients and families, it offers clarity and an opportunity to fight the disease at its earliest stage. For researchers, it provides an indispensable tool to accelerate therapy development. The dissemination of these findings in high-impact scientific publications, such as the Nature Portfolio journals, available through online-access subscription services or in traditional print issues (with costs that may vary based on factors like local taxes), ensures this knowledge spreads rapidly through the scientific community. This virtuous cycle of better diagnosis leading to better trials and, finally, to effective treatments is now within sight.
Conclusion: Solidifying αSyn-SAA’s Role as a Game-Changer
The arrival of Alpha-Synuclein Seed Amplification Assays marks a watershed moment in the history of Parkinson Disease. It is the culmination of decades of research, translating a deep understanding of protein misfolding into a clinical tool with transformative power. This technology is not merely an incremental advance; it is a disruptive innovation that fundamentally changes the rules of the game.
Recapping the Transformative Power of αSyn-SAA
The assay’s impact is threefold. First, it revolutionizes diagnosis by replacing subjective clinical assessment with an objective, biological confirmation of the core pathology, enabling detection at the earliest prodromal stage. Second, it transforms clinical trials by enabling precise patient selection and providing a potential means of objectively measuring therapeutic efficacy. Third, it provides immense clarity and hope to patients, ending diagnostic uncertainty and opening the door to proactive disease management.
Ushering in a New Era of Precision Medicine for Parkinson’s
With αSyn-SAA, we are moving away from a one-size-fits-all approach to Parkinson’s and entering an era of precision medicine. The ability to define the disease based on its underlying biology allows us to target the right pathology in the right patient at the right time. As the assay becomes more accessible through less invasive sampling and is integrated with other genetic and molecular biomarkers, it will become the cornerstone of a new clinical paradigm focused not just on managing symptoms but on intervening early to preserve neurological function, offering a tangible horizon of hope for millions worldwide.
2 Comments
-
Nanu Carmago
I battled advanced Parkinson’s disease for 8 years, starting at age 56. My symptoms included tremors, muscle stiffness, slow movement, and severe balance issues. Despite trying many conventional treatments, nothing brought lasting relief. Then I discovered the P-D treatment program from Dr. Madida Sam at Earth Cure Herbal Clinic I found on Youtube with a search of Dr Madida Sam on YouTube. I found their contact at www. earthcureherbalclinic. com and reached out via email at info(AT)earthcureherbalclinic. com. Within 6 months of starting the program, my symptoms were completely gone, something I never thought possible. I’m incredibly grateful to Dr. Madida Sam and his team for helping me regain my life. If you’re struggling with chronic illness and looking for an alternative approach, I highly recommend giving this a try. It truly changed everything for me..



Nanu Carmago
I battled advanced Parkinson’s disease for 8 years, starting at age 56. My symptoms included tremors, muscle stiffness, slow movement, and severe balance issues. Despite trying many conventional treatments, nothing brought lasting relief. Then I discovered the P-D treatment program from Dr. Madida Sam at Earth Cure Herbal Clinic I found on Youtube with a search of Dr Madida Sam on YouTube. I found their contact at www. earthcureherbalclinic. com and reached out via email at info(AT)earthcureherbalclinic. com. Within 6 months of starting the program, my symptoms were completely gone, something I never thought possible. I’m incredibly grateful to Dr. Madida Sam and his team for helping me regain my life. If you’re struggling with chronic illness and looking for an alternative approach, I highly recommend giving this a try. It truly changed everything for me.