
Huntington’s Disease
Huntington’s disease (HD) is a devastating, inherited neurodegenerative disorder that profoundly impacts individuals and their families. It is characterized by a progressive decline in motor control, cognitive function, and psychiatric well-being. While its genetic roots are well-understood, the complex interplay of symptoms, cellular damage, and the ongoing search for effective treatments presents a significant challenge. This article aims to demystify Huntington’s disease by exploring its genetic causes, detailing its diverse symptoms, outlining diagnostic approaches, and shedding light on the cutting-edge research paving the way for future therapies and improved care. By fostering a deeper understanding, we can empower patients, families, and the medical community to navigate this complex condition with greater knowledge and hope. Worldwide, the prevalence of Huntington’s disease (HD) was 3.92 cases per 100,000 as of 2022, with higher rates observed in Europe (5.65 per 100,000) and North America (7.43 per 100,000) source text.
What is Huntington’s Disease?
Huntington’s disease is a rare but formidable genetic disorder that leads to the progressive breakdown of nerve cells in the brain. This degeneration affects multiple crucial areas of the brain, resulting in a spectrum of debilitating symptoms. The disease typically manifests in adulthood, though juvenile forms exist, and its progression is relentless, impacting a person’s ability to move, think, and manage their emotions. It is classified as a genetic disorder that follows a specific pattern of inheritance.
The Importance of Understanding HD: Causes, Symptoms, and Research
A comprehensive understanding of Huntington’s disease is paramount for several reasons. Firstly, grasping its causes empowers at-risk individuals and families to make informed decisions regarding genetic testing and family planning. Secondly, recognizing the diverse symptoms is crucial for timely diagnosis, effective management, and providing appropriate support. Finally, staying abreast of current research offers hope by illuminating potential therapeutic avenues and driving the development of treatments that can improve the quality of life for those affected by this challenging genetic disorder.
The Genetic Basis: Understanding the Cause of Huntington’s Disease
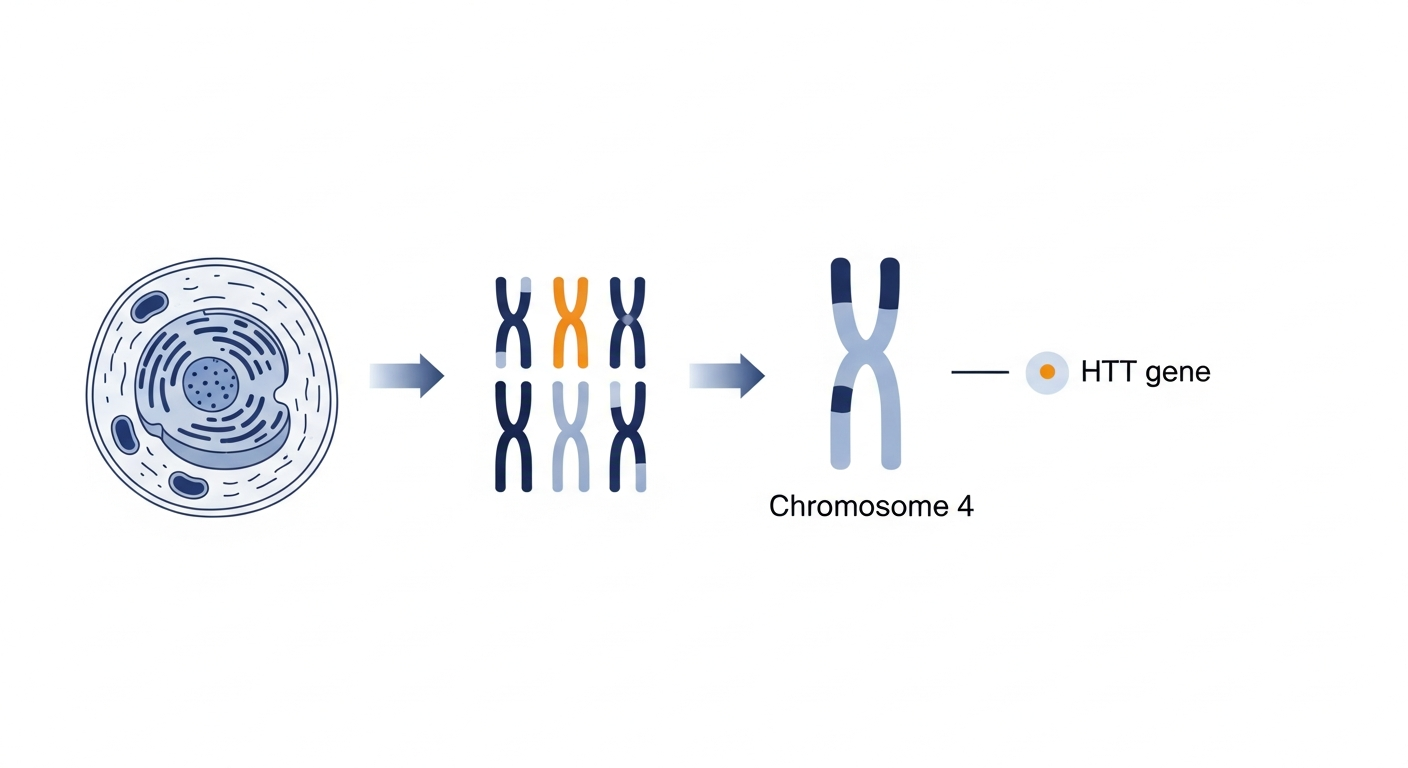 The genetic basis of Huntington’s disease is a mutation in the Huntingtin (HTT) gene, which is located on the short arm of Chromosome 4.
The genetic basis of Huntington’s disease is a mutation in the Huntingtin (HTT) gene, which is located on the short arm of Chromosome 4.
At the heart of Huntington’s disease lies a specific genetic mutation, a flaw that dictates the development and progression of this neurodegenerative condition. Understanding this genetic foundation is key to comprehending the disease’s inheritance pattern and the molecular mechanisms that lead to cellular damage.
The Huntingtin Gene (HTT gene) and Chromosome 4
The root cause of Huntington’s disease resides in a specific gene, the Huntingtin gene, also known as the HTT gene. This gene is located on chromosome 4, one of the 23 pairs of chromosomes that make up the human genome. The Huntingtin gene provides instructions for making a protein called huntingtin. In its healthy form, the huntingtin protein plays a vital role in the normal development and function of nerve cells in the brain.
The Critical Role of CAG Repeats: How the Gene Goes Awry
The problem arises when a section of the Huntingtin gene containing a DNA building block sequence – cytosine, adenine, and guanine (CAG) – is repeated an abnormal number of times. In healthy individuals, this CAG sequence typically repeats between 10 and 35 times. However, in people with Huntington’s disease, this repeat sequence is expanded. The number of CAG repeats generally ranges from 36 to over 100. A greater number of CAG repeats often correlates with an earlier age of onset and potentially a more rapid progression of the disease. Approximately 1 in 400 individuals from the general population carry an expanded CAG repeat associated with Huntington’s disease (≥36 CAG repeats), according to a study that evaluated 7,315 individuals source text.
The Mutant Huntingtin Protein: From Gene Change to Cellular Damage
When the CAG sequence is excessively repeated, it leads to the production of a faulty, longer version of the huntingtin protein, known as the mutant Huntingtin protein. This aberrant protein is toxic to nerve cells, particularly in critical areas of the brain such as the striatum and cerebral cortex. The exact mechanisms by which the mutant protein causes damage are complex and multifaceted, but it is believed to disrupt normal cellular processes, leading to nerve cells malfunctioning and eventually dying.
Autosomal Dominant Inheritance: Understanding the Family Link
Huntington’s disease follows an autosomal dominant inheritance pattern. This means that a person only needs to inherit one copy of the altered Huntingtin gene from either parent to develop the condition. If a parent has Huntington’s disease, each of their children has a 50% chance of inheriting the gene mutation and developing the disease. This inherent family link underscores the importance of genetic testing and genetic counseling for individuals with a known family history of HD.
The Spectrum of Symptoms: How HD Affects the Body and Mind
The hallmark of Huntington’s disease is its insidious progression and the wide-ranging symptoms it encompasses, affecting motor function, cognitive abilities, and emotional well-being. These symptoms typically emerge between the ages of 30 and 50, though variations in age of onset are significant. While the majority of individuals with Huntington’s disease develop motor symptoms in their forties and fifties, approximately 10% experience symptom onset after age 60, and another 10% are affected by Juvenile-onset HD, with symptoms appearing before age 20 source text.
Movement Disorders (Motor Symptoms)
Motor symptoms are often the most outwardly visible signs of Huntington’s disease. The most characteristic is chorea, a series of rapid, jerky, involuntary movements that can appear dance-like. These involuntary movements can affect the face, trunk, and limbs, becoming more pronounced with stress or excitement. Other motor disturbances include:
- Dystonia: Muscle rigidity and stiffness, which can cause abnormal postures and slow, sustained muscle contractions.
- Bradykinesia: Slowness of movement, which can lead to difficulty initiating actions and a reduced range of motion.
- Gait Disturbances: Unsteady walking, balance problems, and an increased risk of falls.
- Speech and Swallowing Difficulties: Problems articulating words clearly and challenges with swallowing food and liquids.
Cognitive Changes and Intellectual Decline
Beyond motor impairments, Huntington’s disease significantly impacts cognitive function. These changes can affect a person’s ability to think, reason, and process information, often preceding or co-occurring with motor symptoms. Key cognitive deficits include:
- Difficulty with executive functions: Problems with planning, organizing, multitasking, and problem-solving.
- Impaired judgment and decision-making: Leading to challenges in daily management and financial affairs.
- Memory issues: While not always the primary deficit, memory recall can become problematic.
- Reduced mental flexibility: Difficulty adapting to new situations or changing routines.
- Speech and language impairments: Affecting comprehension and expression.
Psychiatric Symptoms and Emotional Impact
The psychiatric symptoms associated with Huntington’s disease can be profound and significantly affect a person’s quality of life and relationships. These often emerge early and can be particularly distressing for both the individual and their family. Common psychiatric manifestations include:
- Depression: Persistent sadness, loss of interest, and feelings of hopelessness are very common. Among Huntington’s disease patients, 55.9% reported experiencing stigma, and 37.4% exhibited depressive symptoms, both significantly contributing to a lower quality of life source text.
- Anxiety and irritability: Increased nervousness, agitation, and a shorter temper.
- Apathy and lack of motivation: Loss of interest in activities and social engagement.
- Obsessive-compulsive behaviors: Repetitive thoughts or actions.
- Psychosis: In some cases, hallucinations or delusions may occur.
Disease Progression and Variability
Huntington’s disease is a progressive disorder, meaning its symptoms worsen over time. People with Huntington’s disease typically live for about 15 to 20 years after the onset of symptoms, with the rate of progression varying among individuals source text. The specific combination and severity of motor, cognitive, and psychiatric symptoms can differ significantly from person to person, influenced by factors such as the number of CAG repeats and genetic modifiers. This variability makes predicting individual disease trajectories challenging.
Diagnosis: Identifying Huntington’s Disease
Diagnosing Huntington’s disease involves a comprehensive evaluation that combines clinical observation, patient history, neurological assessment, and definitive genetic testing. Early and accurate diagnosis is critical for initiating appropriate care and support.
Clinical Evaluation: The Role of Physicians and Neurologists
The diagnostic journey typically begins with a visit to physicians, often starting with a primary care doctor who may refer the patient to a neurologist or a movement disorder specialist. These specialists will conduct a thorough neurological examination, observing for characteristic motor signs such as chorea, rigidity, and balance issues. They will also inquire about family history of the disease and assess cognitive and psychiatric symptoms. This clinical assessment helps physicians to form a preliminary diagnosis and rule out other conditions.
The Definitive Genetic Test: A Blood Test for CAG Repeats
The definitive diagnosis of Huntington’s disease is confirmed through genetic testing. This involves a simple blood test that analyzes a sample of DNA to count the number of CAG repeats in the Huntingtin gene. The results clearly indicate whether the individual carries the mutation associated with HD. For those at risk due to family history, predictive genetic testing can provide certainty. However, the decision to undergo genetic testing is significant, and professional genetic counseling is strongly recommended before and after testing to discuss implications, results, and emotional support. Only 12% to 17% of adults at risk for Huntington’s disease in North America and Europe currently pursue predictive genetic testing source text.
Brain Imaging: Observing Changes in the Brain
While not diagnostic on its own, brain imaging techniques such as Magnetic Resonance Imaging (MRI) or Computed Tomography (CT) scans can provide valuable supportive evidence. These scans can reveal characteristic patterns of brain atrophy, particularly in the basal ganglia (including the caudate nucleus and putamen), which are key areas affected by Huntington’s disease. These imaging findings can help physicians confirm the presence of neurodegeneration consistent with HD and differentiate it from other neurological disorders.
Differential Diagnosis: Distinguishing HD from Other Neurodegenerative Disorders
The symptoms of Huntington’s disease can overlap with those of other neurological conditions, making differential diagnosis essential. Conditions such as Parkinson’s disease, Wilson’s disease, Huntington-like syndromes, and certain forms of dementia can present with similar motor, cognitive, or psychiatric disturbances. A comprehensive clinical evaluation, detailed family history, and definitive genetic test are crucial for accurately distinguishing Huntington’s disease from these other neurodegenerative disorders.
Decoding Neurodegeneration: The Cellular and Molecular Mechanisms
Understanding the precise ways Huntington’s disease damages the brain at a cellular and molecular level is crucial for developing targeted therapies. This involves delving into the toxic cascade initiated by the mutant Huntingtin protein.
The Toxic Role of the Mutant Huntingtin Protein
The mutant Huntingtin protein is the central culprit in HD pathogenesis. Once produced, this misfolded protein tends to aggregate, forming clumps within and around nerve cells. These aggregates can disrupt normal cellular functions in several ways, including interfering with gene expression, protein degradation pathways, and intracellular transport. The accumulation of these toxic protein species triggers a cascade of cellular damage that ultimately leads to neuron death.
Impact on Brain Structures and Neurons
The progressive loss of nerve cells (neurons) in Huntington’s disease primarily affects the striatum, a crucial component of the basal ganglia involved in motor control, learning, and habit formation. The cerebral cortex, responsible for higher-level cognitive functions, is also significantly impacted. The degeneration of these brain regions explains the characteristic motor, cognitive, and psychiatric symptoms seen in HD patients. The selective vulnerability of these neurons to the mutant Huntingtin protein remains an active area of research.
Emerging Cellular Pathways and Dysfunctions
Beyond direct protein aggregation, research is uncovering a host of other cellular dysfunctions contributing to HD pathogenesis. These include mitochondrial dysfunction (impairment of cellular energy production), oxidative stress (damage caused by unstable molecules called free radicals), impaired protein clearance mechanisms, and disruptions in nerve cell communication. Understanding these interconnected pathways offers multiple targets for therapeutic intervention.
Current Management and Supportive Care
While a cure for Huntington’s disease remains elusive, significant progress has been made in managing its diverse symptoms and providing comprehensive supportive care. The goal is to maximize function, maintain quality of life, and provide comfort to individuals and their families. The global Huntington’s Disease Treatment Market was valued at USD 1.23 billion in 2023 and is projected to reach USD 3.29 billion by 2033, demonstrating a Compound Annual Growth Rate (CAGR) of 10.34% source text.
Symptom Management with Medicines
Medicines play a vital role in managing the symptoms of Huntington’s disease. For the involuntary movements characteristic of chorea, drugs like tetrabenazine and deutetrabenazine are often prescribed. These medicines work by depleting dopamine in the brain, which can help to reduce the severity of jerky movements. Other medicines are used to manage psychiatric symptoms, including antidepressants for depression and anxiety, and antipsychotics for irritability or psychosis. The choice and dosage of medicines are tailored to each individual’s specific needs and are managed by physicians.
Therapeutic Interventions: Maximizing Function and Quality of Life
Beyond pharmacotherapy, a multidisciplinary approach involving various therapeutic interventions is crucial. Speech therapy can help with communication and swallowing difficulties, while physical therapy can improve strength, balance, and mobility, reducing the risk of falls. Occupational therapy focuses on adapting daily living activities to help individuals maintain independence and manage tasks more effectively. Nutritional support is also vital, particularly for individuals experiencing swallowing difficulties or weight loss.
Holistic Care and Long Term Care Considerations
Holistic care for Huntington’s disease extends beyond medical management to encompass emotional, social, and practical support. Family involvement is paramount, but caring for individuals with HD presents significant challenges. A systematic review of studies involving 569 caregivers concluded that caring for individuals with HD involves a distinct and multifaceted burden, significantly impacted by the severity of patient symptoms and compromised caregiver mental health source text. Therefore, long term care planning, including considerations for in-home support, assisted living, or specialized care facilities, is essential. Palliative care specialists can also play a vital role in managing complex symptoms and supporting both patients and their families throughout the disease trajectory.
The Frontier of Research: New Hope and Future Therapies
The landscape of Huntington’s disease research is dynamic and filled with promise, offering new avenues for developing disease-modifying treatments. As of March 2024, numerous registered and ongoing clinical trials are actively investigating new treatments for Huntington’s disease, including programs from VICO Therapeutics, uniQure, and PTC Therapeutics source text.
Targeting the Gene: Gene Silencing and Gene Editing Strategies
A major focus of current research is on directly targeting the genetic root of HD. Gene silencing therapies, such as antisense oligonucleotides (ASOs) and small interfering RNAs (siRNAs), aim to reduce the production of the toxic mutant Huntingtin protein. These therapies are designed to interfere with the messenger RNA (mRNA) that carries the genetic code from the gene to the protein-making machinery of the cell. Gene editing technologies, like CRISPR-Cas9, are also being explored for their potential to correct the mutation in the Huntingtin gene itself.
Understanding and Monitoring Disease Progression
Developing reliable biomarkers to monitor disease progression and treatment response is another critical area of research. Advances in imaging techniques, fluid biomarkers (found in cerebrospinal fluid or blood), and digital phenotyping (using wearable sensors to track movement and behavior) are helping researchers better understand the subtle changes that occur in the brain and body over time. This is crucial for evaluating the efficacy of new therapies in clinical trials.
Emerging Therapeutic Avenues
Beyond gene-targeting approaches, researchers are exploring other promising strategies. These include developing drugs that prevent the aggregation of the mutant Huntingtin protein, compounds that protect nerve cells from damage (neuroprotective agents), and therapies aimed at restoring normal cellular functions that are disrupted by the disease. Understanding the intricate molecular pathways involved in HD pathogenesis continues to reveal new targets for innovative treatments.
Conclusion
Huntington’s disease is a complex and profoundly challenging genetic disorder that impacts multiple facets of an individual’s life. From its origins in a specific mutation on chromosome 4 and the Huntingtin gene, to the wide-ranging symptoms affecting movement, cognitive function, and emotional health, HD demands a thorough understanding. The progressive nature of the disease, characterized by the degeneration of nerve cells in the brain, underscores the urgency for effective interventions.
While no cure currently exists, significant strides are being made. The diagnostic process, relying on clinical assessment by physicians and definitive genetic testing, allows for early identification. Current management focuses on alleviating symptoms through medicines like tetrabenazine, alongside vital therapies and supportive care aimed at enhancing quality of life. Crucially, the frontier of research, particularly in gene silencing and other targeted therapies, offers a beacon of hope for future breakthroughs.
Navigating life with Huntington’s disease requires a holistic approach, encompassing medical management, emotional support, and careful long term care planning for both patients and their families. The significant burden on caregivers highlights the need for robust support systems, as caring for individuals with HD is complex and multifaceted. Despite the challenges, the active pursuit of knowledge and innovative treatments fuels optimism for a future where Huntington’s disease can be more effectively managed and potentially overcome.
.



Leave a Reply