Cytokine testing services: How to choose the right CRO for neurotherapeutics
Cytokine testing services are all about measuring tiny "messenger" proteins, called cytokines, that orchestrate our immune responses and inflammation. By measuring the levels of these proteins in samples like blood or cerebrospinal fluid (CSF), these services give us a direct window into the body's inflammatory state. This is especially vital for cracking the code of neurological diseases.
The Growing Role of Cytokine Testing in Neurology

Think of the central nervous system (CNS) as a highly secured fortress, walled off by the blood-brain barrier. For a long time, the scientific community believed the immune activity inside this fortress was isolated from the rest of the body. We now know that’s not the case at all.
There’s a constant, dynamic conversation happening between the brain and the immune system, and cytokines are the language they speak.
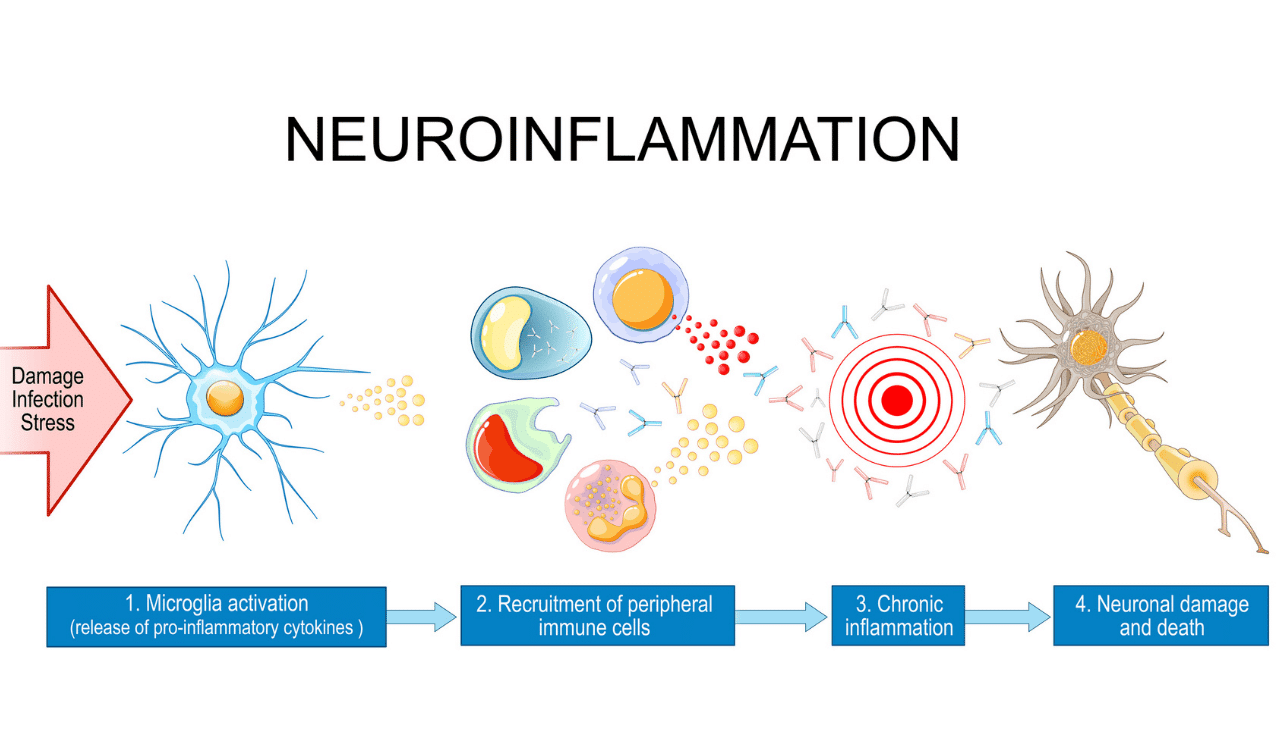
These small proteins are the master communicators in the body's inflammatory network. When everything is working correctly, they coordinate responses to injury and infection, then give the "all-clear" for repair and a return to balance. But in neurodegenerative diseases like Alzheimer's, Parkinson's, and multiple sclerosis, this communication network goes haywire.
Decoding Neuroinflammatory Signals
The cytokine conversation gets completely out of whack. Certain proteins, known as pro-inflammatory cytokines, get stuck in an "on" position, leading to a state of chronic, smoldering inflammation that is toxic to neurons.
At the same time, the anti-inflammatory cytokines that are supposed to put out this fire can become suppressed or simply overwhelmed. This persistent neuroinflammation isn't just a symptom; we now recognize it as a core driver of disease progression. It’s the slow-burning fire that accelerates neuronal damage and cognitive decline.
Measuring cytokines provides a direct readout of this inflammatory activity. It transforms abstract concepts like "neuroinflammation" into concrete, quantifiable data points that can be tracked over time.
This is precisely what makes cytokine testing services so indispensable in modern neurotherapeutic development. They give researchers the ability to:
- Understand Disease Mechanisms: Pinpoint exactly which inflammatory pathways are firing in a specific disease.
- Stratify Patient Populations: Identify patient subgroups based on their unique inflammatory profiles, which helps design smarter, more targeted clinical trials.
- Assess Drug Efficacy: Get a clear answer on whether a new therapy is actually dampening harmful inflammation or promoting repair.
The field's importance is mirrored by its explosive growth. The global cytokine market was valued at USD 95.70 billion in 2024 and is projected to skyrocket to USD 172.36 billion by 2033. This surge underscores the growing reliance on cytokine analysis to develop the next generation of treatments. You can dig into the full market projections about cytokine market growth for more details.
Key Cytokines in Neuroinflammation and Their Functions
While hundreds of cytokines exist, a core panel is frequently analyzed in neurological studies because their roles are well-established. Understanding this "shortlist" is key to making sense of the data that cytokine testing services provide.
Here's a quick look at some of the major players and what they do.
| Cytokine | Primary Role | Associated Neurological Conditions |
|---|---|---|
| TNF-α | A potent pro-inflammatory signal; a primary driver of inflammation. | Alzheimer's Disease, Multiple Sclerosis, Stroke |
| IL-6 | Pro-inflammatory; heavily involved in the acute phase response. | Parkinson's Disease, Traumatic Brain Injury |
| IL-1β | A key initiator of the entire inflammatory cascade. | Epilepsy, Alzheimer's Disease |
| IL-10 | A major anti-inflammatory cytokine; works to suppress immune responses. | Multiple Sclerosis, Neuroinflammation |
| IFN-γ | Pro-inflammatory; critical for antiviral defense but can drive autoimmunity. | Viral Encephalitis, Autoimmune Disorders |
By monitoring these and other relevant markers, drug developers can paint a detailed picture of a disease's inflammatory landscape. This moves research beyond just observing symptoms and toward targeting the fundamental biological processes that cause neuronal decline, paving the way for more effective, data-driven therapeutic strategies.
Picking the right cytokine assay is a bit like a neurosurgeon choosing a scalpel—the wrong tool can jeopardize the whole operation. The world of cytokine testing services is packed with different platforms, and each comes with its own set of trade-offs. The key is to match the tool to the specific job you need to get done, balancing sensitivity, throughput, multiplexing power, and, of course, budget.
Your starting point should always be the central question of your study. Are you in an early discovery phase, just trying to get a lay of the land to see which inflammatory pathways are lighting up? Or are you much further along, needing to prove that your drug affects a specific, low-level cytokine in a tricky sample like cerebrospinal fluid (CSF)?
The answer completely changes the technology you should reach for. A broad, exploratory tool is perfect for that initial wide-net screening, but you’ll need a highly specialized, ultra-sensitive instrument when it's time for precise, targeted confirmation.
Traditional vs. High-Throughput Platforms
For decades, the Enzyme-Linked Immunosorbent Assay (ELISA) has been the workhorse for measuring a single protein. Think of an ELISA as a trusty, high-powered magnifying glass—it’s fantastic for looking at one thing very, very closely. It’s reliable, relatively inexpensive, and everyone understands how it works, making it a go-to choice for validating a key finding or tracking a single, well-established biomarker. If you're interested in the nuts and bolts of this foundational method, you can learn more about what goes into high-quality ELISA kit production.
But modern neurotherapeutics often demand a much bigger picture. This is where multiplex platforms change the game, letting you measure dozens of cytokines at once from a single, precious sample.
Multiplexing is like swapping that single magnifying glass for a high-resolution satellite map. Instead of zeroing in on one house, you suddenly see the entire neighborhood, how the streets connect, and the flow of traffic. It gives you a complete picture of the inflammatory landscape.
Some of the most common multiplex technologies include:
- Luminex/xMAP: This platform from Luminex Corporation uses tiny, color-coded beads, each coated with an antibody for a different cytokine. It's a fantastic screening tool that lets you measure up to 80 or more analytes simultaneously, perfect for broad discovery panels.
- Meso Scale Discovery (MSD): This technology from Meso Scale Diagnostics uses electrochemiluminescence to deliver impressive sensitivity and a wide dynamic range. Offering both pre-made and custom panels, MSD hits a sweet spot between multiplexing capability and high-performance data.
While incredibly powerful, these platforms need careful planning. The more analytes you cram into a single panel, the higher the risk of cross-reactivity or other interferences that can create noise and muddy your data.
Pushing the Limits with Ultra-Sensitive Assays
So, what do you do when cytokine levels are just too low for even high-quality ELISAs or multiplex assays to see? This is a constant headache in neurology, especially when you're hunting for CNS-specific signals that are heavily diluted in blood or CSF.
This is where ultra-sensitive assays become absolutely critical. Technologies like Single Molecule Array (Simoa) from Quanterix can detect proteins down to the femtogram/mL level. That’s thousands of times more sensitive than a standard ELISA. It’s like moving from a light microscope to a powerful electron microscope, allowing you to see individual molecules that were completely invisible before.
This kind of sensitivity is a game-changer for neuroinflammation research. It finally allows for the reliable measurement of crucial biomarkers like IL-6, TNF-α, and neurofilament light (NfL) directly from a simple blood draw, opening up entirely new possibilities.
A Practical Comparison of Cytokine Assay Technologies
Choosing the right platform is always a balancing act between your scientific goals, your sample availability, and your budget. This table offers a straightforward comparison to help you match the technology to your project's specific needs.
| Platform | Sensitivity | Throughput | Best Use Case | Key Limitation |
|---|---|---|---|---|
| ELISA | Moderate | Low (single-plex) | Validating a specific, known biomarker. | Inefficient for screening multiple analytes. |
| Luminex | Moderate | High (up to 80-plex) | Broad, early-stage discovery screening. | Potential for cross-reactivity in large panels. |
| MSD | High | Medium (up to 10-plex/well) | Targeted panels requiring high sensitivity and dynamic range. | Higher cost per sample compared to Luminex. |
| Simoa | Ultra-High | Low (up to 4-plex) | Detecting very low-abundance biomarkers in blood/CSF. | Limited multiplexing capability and higher cost. |
Ultimately, the best cytokine testing services aren't just about having the latest machines. They're about offering a range of these platforms backed by the scientific expertise to help you select and validate the optimal tool for your unique neurotherapeutic program.
Get the Pre-Analytical Steps Right, or Nothing Else Matters
You can have the most advanced cytokine testing service in the world, but it’s completely useless if your samples are compromised from the very beginning. Flawless pre-analytical handling—every single step taken before a sample hits the assay plate—is the absolute bedrock of a successful biomarker strategy. Even the smallest missteps can inject huge variability into your results, leading to skewed data and, ultimately, dead-end conclusions.
Think of it this way: your sample is a pristine recording of a crucial biological conversation happening inside a patient. The collection, processing, and storage steps are the sound engineering. If your microphone is faulty or the recording is handled poorly, you’ll just get static and noise, completely drowning out the original message. A single, unnecessary freeze-thaw cycle can wreck sensitive cytokines, permanently corrupting the data you’re trying to capture.
Standardizing Sample Collection and Handling
To make sure every "recording" is crystal clear, you need rigorous and standardized protocols. These are non-negotiable, and they start the second the sample is drawn.
Here are the key best practices that can make or break your study:
- Consistent Collection Tubes: The type of tube you use for a blood draw—whether it's EDTA, heparin, or a serum separator—can dramatically change measured cytokine levels. Always use the exact same tube type for every subject and every time point in a study. Consistency here is key to eliminating this as a variable.
- Controlled Processing Times: The clock starts ticking the moment blood is drawn. The time it takes to get that sample to a centrifuge to separate plasma or serum is critical. If left sitting at room temperature, blood cells can either release or degrade cytokines. A strict protocol, like processing every single sample within 60 minutes of collection, is essential.
- Minimizing Freeze-Thaw Cycles: This is one of the most common—and most damaging—sources of error. Every time a sample is frozen and thawed, fragile protein structures can be destroyed. The best practice is to aliquot samples into single-use volumes right after the first processing step. That way, you avoid the need for repeated freeze-thaw cycles altogether.
This simple flowchart can help you connect your study goals—whether you're in a broad discovery phase or a targeted confirmation stage—with the right analytical approach from day one.

As the diagram shows, the fundamental question you're asking should dictate your tools. Are you casting a wide net for new discoveries, or are you using a precision instrument to confirm a specific hypothesis?
Navigating GLP and CLIA Regulatory Frameworks
Beyond just handling protocols, the regulatory environment your cytokine testing services operate in is just as critical for ensuring your data holds up to scrutiny. The two major standards you'll run into are Good Laboratory Practice (GLP) and the Clinical Laboratory Improvement Amendments (CLIA).
They might sound similar, but they serve very different purposes. Knowing the difference is absolutely vital for making sure your biomarker strategy aligns with your program's ultimate goals, whether that's an internal go/no-go decision or a formal submission to the FDA.
GLP and CLIA aren't just bureaucratic hoops to jump through. They are robust frameworks designed to ensure your data is solid, reproducible, and fit for its intended purpose. The right choice depends entirely on how your data will be used.
Let's break down what each standard actually means for your project:
Good Laboratory Practice (GLP)
- Focus: GLP is all about non-clinical lab studies that will be used to support applications to regulatory agencies like the FDA.
- Purpose: It’s designed to guarantee the uniformity, consistency, reliability, and quality of preclinical safety data. The framework governs the entire lifecycle of a study—from planning and execution to monitoring, recording, and archiving.
- When It's Needed: You need a GLP-compliant lab when your cytokine data is going to be part of a formal submission package for a new drug or therapeutic.
Clinical Laboratory Improvement Amendments (CLIA)
- Focus: CLIA regulates all lab testing performed on human samples in the U.S. that will be used for diagnosis, prevention, or treatment of disease.
- Purpose: Its goal is to ensure the accuracy, reliability, and timeliness of patient test results, no matter where the test is performed.
- When It's Needed: CLIA certification is mandatory if your cytokine results will be used to make decisions about individual patient care. This includes things like stratifying patients for a clinical trial or developing a companion diagnostic.
Choosing a CRO that has deep expertise in both GLP and CLIA environments is a huge advantage. It means they have the quality systems and regulatory know-how to support your program from early discovery all the way through to clinical application. It’s your guarantee that the data you generate isn’t just scientifically interesting—it’s defensible.
Weaving the Whole Story with Integrated Biomarkers

Cytokine data gives you a powerful snapshot of neuroinflammation, but inflammation is just one chapter in the complex story of neurological disease. To really grasp what’s happening in the brain, we have to look beyond a single data stream and build a more complete, integrated picture.
Think of it like being a detective at a crime scene. A single clue—the cytokine signal—tells you something is wrong. But it doesn't tell you the whole story. You need to combine it with other evidence to understand what happened and why. For neurotherapeutics, this means pairing cytokine analysis with other classes of biomarkers that measure the downstream consequences of that inflammation.
This systems-biology view creates a multi-dimensional picture, allowing you to connect the dots between an inflammatory flare-up and its real-world impact on neuronal health. It’s how we move from simply measuring inflammation to practicing true precision neurology.
Correlating Inflammation with Neuronal Injury
One of the most powerful strategies is to directly correlate pro-inflammatory cytokine levels with markers of neuronal damage. This is how you confirm that the "smoke" (inflammation) is actually coming from a "fire" (neuronal injury). It’s a simple but profound step.
A classic example involves measuring two key proteins in parallel:
- Interleukin-6 (IL-6): A well-known pro-inflammatory cytokine that often signals an active immune response within the central nervous system.
- Neurofilament Light Chain (NfL): A structural protein that lives inside neurons. When neurons are damaged or die, NfL leaks out into the cerebrospinal fluid and, in smaller amounts, into the bloodstream.
If your study shows a new drug lowers IL-6, that’s a promising start. But if it shows the drug also lowers NfL levels in the same patients, the evidence becomes far more compelling. You’ve just demonstrated that by dialing down inflammation, your drug is actively protecting neurons from being destroyed.
This kind of integrated analysis turns a simple pharmacodynamic (PD) marker into a powerful tool that clarifies your drug's mechanism of action and builds a much stronger case for its clinical benefit. For a deeper look at this landscape, you can explore the broader field of neurological biomarkers research services and see how these pieces fit together.
A Multi-Biomarker Approach in Practice
Integrating different biomarker classes allows you to ask much more sophisticated questions and de-risk your clinical program. It gives you a richer, more contextualized view of disease pathology and how a patient is responding to treatment.
By combining markers of inflammation, cell stress, and specific neuronal damage, you can create a detailed signature of a drug's effect. This moves you from a simple "on/off" switch to a nuanced understanding of pathway engagement.
Here’s how you can weave different biomarker types together to tell a more complete story:
- Inflammatory Markers (e.g., TNF-α, IL-1β): Pinpoint the specific inflammatory pathways being activated or suppressed.
- Neuronal Injury Markers (e.g., NfL, Tau): Directly measure the extent of damage to axons and neurons.
- Synaptic Health Markers (e.g., SNAP-25): Assess the impact on synaptic function and plasticity, which is closely tied to cognitive performance.
- Cellular Pathway Markers (e.g., LC3 for autophagy): Reveal how a drug impacts core cellular processes like waste clearance or metabolic function.
Imagine you're running a clinical trial for a new Alzheimer's drug. By using an integrated biomarker panel, you could show that your drug not only reduces key cytokines but also lowers levels of pathogenic p-tau and stabilizes synaptic protein levels. This multi-pronged evidence provides a much stronger biological rationale for any observed cognitive improvements and significantly strengthens a regulatory submission.
How to Select the Right Cytokine Testing Partner
Choosing a contract research organization (CRO) for your cytokine testing services is one of the most consequential decisions you'll make in your neurotherapeutic development program. This isn’t just about outsourcing a task; it's about forming a partnership that can either de-risk your clinical trial or introduce fatal flaws into your data. A low-cost quote from a generic provider is meaningless if they lack the specialized expertise to deliver reliable, context-rich results.
Your selection process should feel more like hiring a key scientific collaborator than just procuring a service. The right partner becomes an extension of your team, bringing deep experience that helps you navigate the complexities of biomarker analysis. They should not only execute the assays flawlessly but also provide consultative support to help you interpret the results and make sound decisions.
Beyond the Price Tag: Scientific Expertise
The first and most critical filter is scientific expertise—specifically in neurology. A CRO that primarily runs oncology or immunology panels might have the right equipment, but they won't have the nuanced understanding of neuroinflammation required for your program. The CNS is a unique biological environment, and interpreting cytokine signals from samples like CSF or neuron-derived exosomes requires specialized knowledge.
Your vetting process should include direct conversations with the scientists who will be overseeing your project. Ask them pointed questions about their experience with your specific disease indication and sample types.
A true partner doesn't just deliver a spreadsheet of data. They help you understand what that data means in the context of neurodegenerative disease, guiding you from raw numbers to actionable biological insights.
This level of expertise is what separates a mere vendor from a strategic asset. You need a team that understands the challenges of low-abundance analytes in neurological samples and has proven solutions.
A Checklist for Vetting Potential CROs
To ensure you're making a robust choice, use a structured evaluation process. A comprehensive checklist helps you compare partners on the factors that truly matter for the success of your cytokine testing services.
Platform Versatility: Do they offer a range of platforms (e.g., ELISA, Luminex, MSD, Simoa) and have the expertise to help you select the right one for your study's specific goals?
Sample Type Experience: Have they demonstrated success working with your exact sample matrices, whether it’s plasma, serum, CSF, or more specialized preparations like neuron-derived exosomes? Ask for validation data specific to these sample types.
Quality and Regulatory Compliance: Does the lab hold the necessary certifications, such as GLP for regulatory-track studies or CLIA for patient-facing diagnostics? A strong quality management system is non-negotiable.
Data Analysis and Support: Do they provide comprehensive data analysis, including statistical support and interpretation? Can they help you integrate cytokine data with other biomarkers?
For teams exploring how functional assays can complement cytokine measurements, understanding a CRO's capabilities in other areas is also crucial. For example, knowing their approach to cell-based assay services can reveal their broader scientific depth and ability to support integrated biomarker strategies.
Ultimately, selecting the right partner comes down to trust and demonstrated capability. A thorough vetting process that goes far beyond a simple cost comparison will pay dividends, ensuring the data you generate is reliable, defensible, and capable of driving your neurotherapeutic program forward with confidence.
The Future of Neurological Cytokine Analysis
The world of neurological biomarker analysis never sits still, and cytokine testing is certainly no exception. As we look ahead, a few exciting trends are poised to dramatically deepen our grasp of neuroinflammation, helping us design the next wave of precision therapies. These new approaches are moving us beyond simply asking how much of a cytokine is floating around and toward understanding where it’s coming from and how it's actually behaving.
One of the biggest game-changers is the shift toward spatial proteomics. Traditional multiplex assays are powerful, no doubt, but they give you an average reading from a blended sample—it's like grinding up an entire city block to analyze its chemistry. Spatial proteomics, on the other hand, is like having a GPS-enabled map that shows you exactly which cells are making which cytokines and where those signals are being sent within the brain's complex landscape.
This gives researchers a stunningly detailed picture of the inflammatory microenvironment, revealing how immune cells, neurons, and glial cells are all talking to each other in real-time.
The Rise of AI and Functional Assays
With these new, richer datasets, Artificial Intelligence (AI) and machine learning are quickly becoming essential. AI algorithms can dig through enormous amounts of cytokine data—alongside other biomarker and clinical information—to spot subtle patterns a human researcher would almost certainly miss. These algorithms can uncover complex inflammatory signatures that predict how a disease might progress or whether a patient is likely to respond to a particular drug.
Instead of searching for a single "smoking gun" biomarker, AI lets us find an entire "fingerprint" of inflammation. This multi-analyte signature gives us a much more stable and reliable signal for making tough clinical calls.
But it’s not just about measuring protein levels anymore. The field is also leaning heavily into functional cell-based assays. These methods, like the Mixed Lymphocyte Reaction (MLR), test what a drug candidate actually does to immune cell behavior. Researchers can see firsthand if a compound shuts down T-cell proliferation or changes the specific mix of cytokines being secreted, giving a direct readout of a drug's immunomodulatory muscle.
Specialized Therapeutics and Advanced Analysis
We're also seeing the rise of highly specialized immunocytokines, which are cleverly engineered proteins that fuse a cytokine with an antibody. This allows a potent signal to be delivered directly to a specific target cell, avoiding off-target effects. This area is attracting massive investment, which speaks to the broader trend of harnessing cytokine biology as a direct therapeutic strategy.
In fact, the immunocytokines market, valued at USD 847 million in 2023, is forecast to hit USD 5,374 million by 2030. That’s a staggering CAGR of 37.3%. This explosive growth underscores the incredible promise of targeted, cytokine-based treatments. You can dig into the complete immunocytokine market forecast to see the full scope of these innovations.
Taken together, these trends paint a clear picture of how cytokine testing services are maturing. We’re moving from providing simple measurements to delivering deep, actionable insights. By integrating spatial context, AI-driven pattern recognition, and functional readouts, the next wave of cytokine analysis will be a critical partner in developing safer, more effective drugs for neurological disorders.
Common Questions About Cytokine Testing Services
When you're mapping out a neurotherapeutic study, cytokine testing can seem like a complex piece of the puzzle. We hear a lot of the same practical questions from research teams about sample logistics, panel design, and how to get the most relevant data for neurological research. Getting these details right is the bedrock of a solid biomarker strategy.
Let’s tackle some of the most common questions we get.
How Much Sample Do I Need?
This is easily one of the first questions everyone asks, and the answer is usually a relief. Modern multiplex assays, like Luminex or MSD, are incredibly efficient.
Most platforms only need about 25-50 µL of plasma, serum, or CSF for each run. This tiny volume means you can run extensive cytokine panels without placing a heavy burden on your study participants, which is a huge win for everyone.
Should I Use a Broad or Targeted Panel?
The next big question is always about panel strategy: Do you cast a wide net or zero in on a few key targets? The right answer comes down to what you’re trying to achieve in your study.
Broad Discovery Panels (40+ cytokines): Think of these as your exploratory toolkit. They’re perfect for early-phase studies when you don’t have a specific hypothesis. Casting a wide net like this allows you to screen for unexpected pathway changes and discover novel biomarkers you might have otherwise missed.
Targeted Panels (5-10 cytokines): These are your precision instruments. They shine in later-phase trials where you need to confirm a drug's impact on a known inflammatory pathway. A focused panel is more precise, repeatable, and cost-effective for pharmacodynamic (PD) monitoring.
Why Bother Analyzing Cytokines in Exosomes?
For neurology teams, this question is critical. Why look at cytokines inside exosomes when you can just measure them in whole plasma? The answer is all about cutting through the noise to get a clear, brain-specific signal.
Think of it this way: measuring cytokines in whole plasma is like trying to hear a single conversation in a crowded stadium. The signal you care about—the one from the brain—is drowned out by a cacophony of systemic inflammation from the rest of the body. Analyzing cytokines within neuron-derived exosomes (NDEs) is like having a directional microphone pointed directly at that one conversation.
By isolating NDEs first, cytokine testing services can filter out the systemic "noise" and measure the inflammatory environment directly tied to neurons. This gives you a much cleaner, more relevant picture of what's actually happening in the central nervous system—an absolute necessity for developing drugs that work for neurological diseases.
At NeuroDex Inc, we specialize in isolating neuron-derived exosomes to provide the cleanest, most relevant cytokine data for your neurotherapeutic programs. Our GLP- and CLIA-compliant services help you de-risk clinical trials with actionable, brain-specific biomarker insights. Explore our advanced biomarker platforms to accelerate your research.




Leave a Reply