How Extracellular Vesicles Carry Biomarkers from the Brain to Blood
A Comprehensive Review of Brain-Derived EV Biology, BBB Transit Mechanisms, and Diagnostic Applications
Published March 2026 | Neuroscience & Biomarker Research
Introduction: The Challenge of Reading the Brain Without Opening It
The human brain remains one of the most inaccessible organs in clinical medicine. Unlike blood, liver, or kidney tissue, obtaining a real-time molecular snapshot of neurological activity traditionally required either expensive neuroimaging or the invasive and uncomfortable procedure of a lumbar puncture to sample cerebrospinal fluid (CSF). For patients at risk of Alzheimer’s disease, Parkinson’s disease, traumatic brain injury, or psychiatric conditions, neither option is practical for routine, longitudinal monitoring.
This diagnostic gap is precisely why extracellular vesicles (EVs) — tiny, membrane-bound nanoparticles shed by virtually every cell in the body — have captured the imagination of neuroscientists and clinicians worldwide. When brain cells release EVs, those vesicles carry a molecular fingerprint of their parent cell’s state: proteins, messenger RNAs, microRNAs, lipids, and metabolites that faithfully reflect whether that cell is healthy, stressed, or degenerating. Crucially, a subset of these brain-derived EVs escapes into the bloodstream, where they can be captured and analyzed through a simple blood draw.
This article explores the biology behind how extracellular vesicles originate in the brain, how they traverse the blood–brain barrier (BBB), what biomarkers they carry, and why they represent a breakthrough in the field of liquid biopsy for neurological disease.
What Are Extracellular Vesicles? A Primer
Extracellular vesicles are nanosized, lipid bilayer-enclosed particles released by cells during normal physiological activity and in response to stress or injury. Rather than discarding cellular material, EVs encapsulate and protect it, serving as sophisticated couriers in intercellular communication.
Researchers classify EVs into three main subtypes based on their size and biogenesis:
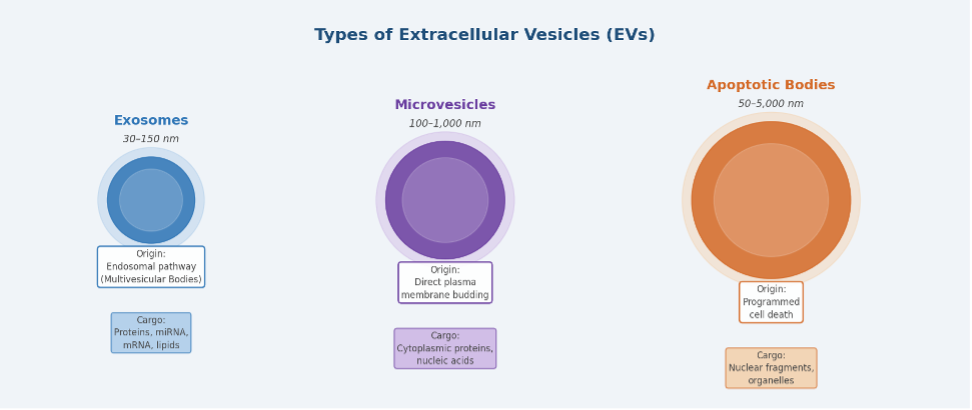
Figure 1. The three main types of extracellular vesicles: exosomes (30–150 nm), microvesicles (100–1,000 nm), and apoptotic bodies (50–5,000 nm), classified by size, biogenesis pathway, and cargo.
Exosomes (30–150 nm): The smallest and most studied subtype, formed within the endosomal pathway. Multivesicular bodies (MVBs) inside the cell fuse with the plasma membrane and release exosomes into the extracellular space. These vesicles carry highly specific cargo — including RNA species and transmembrane proteins — that reflects the transcriptional and translational activity of the parent cell.
Microvesicles (100–1,000 nm): Larger particles that bud directly from the plasma membrane. They tend to carry different cargo profiles compared to exosomes and are released more rapidly in response to cellular activation or damage.
Apoptotic bodies (50–5,000 nm): Released during programmed cell death, these large vesicles contain fragmented nuclear material and organelles. While less useful as diagnostic biomarkers due to their size, they signal active neurodegeneration when elevated in biofluids.
According to Signal Transduction and Targeted Therapy (2024), these vesicles transport diverse cellular components, including proteins, mRNAs, miRNAs, DNA, and lipids across distances, influencing numerous physiological and pathological events. Because EV cargo mirrors the molecular state of its cell of origin, analyzing EVs captured in blood offers a window into tissues that are otherwise inaccessible — including the brain.
Where Do Brain-Derived EVs Come From?
Within the central nervous system (CNS), EVs are produced by every major cell type, each contributing a distinct molecular signature to the circulating EV pool.
Neurons
Neurons are the primary cells of interest for neurological biomarker research. Neuron-derived extracellular vesicles (NDEVs) are released at synapses and along axons, and they carry synapse-specific proteins such as synapsin, PSD-95, GAP43, neurogranin, SNAP25, and synaptotagmin. In disease states, NDEVs also contain pathological proteins — most notably amyloid-beta (Aβ) peptides and hyperphosphorylated tau — that are central to Alzheimer’s disease pathogenesis. As noted in a 2025 review on NDEVs in Developmental Neurobiology, neuron-derived vesicles present in plasma and serum serve as minimally invasive biomarkers, as they carry pathological proteins central to disease.
Astrocytes
Astrocytes, the support cells of the brain, release EVs that carry neuroprotective factors under normal conditions. However, in the context of neuroinflammation or amyloid toxicity, astrocytic EV composition shifts significantly. Research has shown that Aβ peptides and tau proteins alter astrocyte-derived EV content, making these vesicles potential indicators of the neuroinflammatory microenvironment.
Microglia
Microglia are the brain’s resident immune cells and are highly active EV producers, particularly during inflammation. Microglial EVs carry inflammatory cytokines, complement proteins, and miRNAs linked to immune activation. Elevated microglial EV activity has been documented in multiple sclerosis, AD, Parkinson’s disease, and other neuroinflammatory conditions.
Oligodendrocytes
Oligodendrocytes produce myelin and release EVs rich in myelin-related proteins. Amyloid precursor-like protein 1 (APLP1), which has emerged as a novel EV biomarker, is predominantly localized in oligodendrocytes and neurons. A landmark study in Science Advances demonstrated that APLP1-positive EVs in human serum can be unequivocally determined to be of cerebral origin, validated by distinctive small RNA expression patterns and brain-specific surface proteins.
How Do Brain-Derived EVs Cross the Blood–Brain Barrier?
The blood–brain barrier is a highly specialized structure composed of brain microvascular endothelial cells (BMECs), pericytes, and astrocytic endfeet — collectively known as the neurovascular unit. It selectively restricts the passage of molecules from the blood into the brain parenchyma, and vice versa. Understanding how EVs traverse this barrier in the brain-to-blood direction is key to validating them as systemic biomarkers.
A pivotal study published in Pharmaceutics (Banks et al., 2020), examined the BBB pharmacokinetics of 10 different exosome populations. The findings were clear: all exosome populations crossed the BBB, but their rates varied more than tenfold. Crucially, the study also identified a robust brain-to-blood efflux system — meaning that once formed in the brain, EVs are actively transported outward into the peripheral circulation, explaining why CNS-derived vesicles are consistently detectable in blood samples.
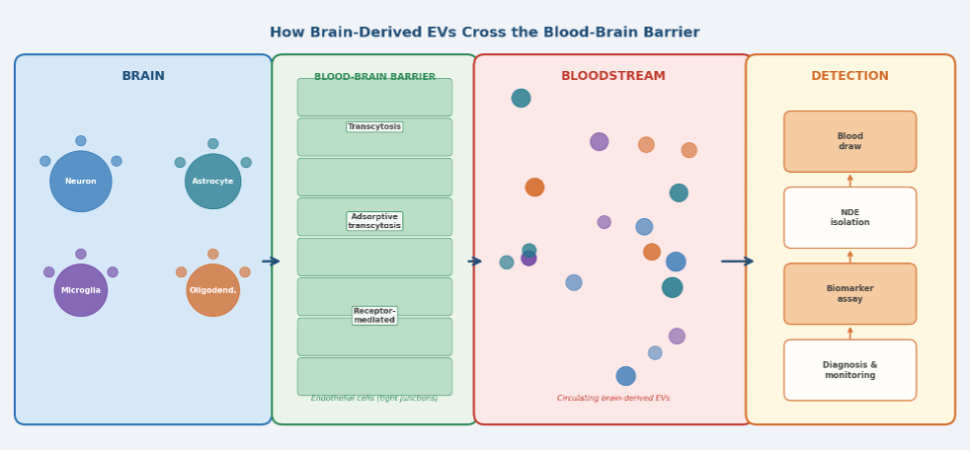
Figure 2. The brain-to-blood transit pathway of extracellular vesicles: EVs released by neurons, astrocytes, microglia, and oligodendrocytes cross the blood–brain barrier via transcytosis and enter the peripheral bloodstream, where they can be isolated and analysed for neurological biomarkers.
Transcytosis: The Primary Transit Mechanism
The dominant mechanism by which EVs transit across the BBB is transcytosis — a multi-step process in which a vesicle is endocytosed on one side of a cellular barrier, transported across the cell interior within endosomes, and then exocytosed on the opposite side. Within this category, several subcategories are relevant:
Adsorptive transcytosis: EVs bind non-specifically to the negatively charged surface of endothelial cells via electrostatic interactions and are then internalized. Evidence from wheatgerm agglutinin (WGA) inhibition studies suggests this pathway handles a significant fraction of EV transit across the BBB.
Receptor-mediated transcytosis: Specific surface proteins on EVs interact with cognate receptors on brain endothelial cells, triggering targeted internalization. The mannose-6-phosphate receptor, for example, has been demonstrated to mediate BBB crossing for specific EV populations.
Micropinocytosis: Small intracellular vacuoles form from plasma membrane invaginations and internalize EVs on the cell surface. This pathway becomes particularly relevant for smaller EV populations and has been visualized in zebrafish BBB models.
BBB Disruption Enhances EV Transit
Under pathological conditions — including neuroinflammation, traumatic brain injury, stroke, and tumor invasion — the integrity of the BBB is compromised. This disruption significantly increases the permeability of the barrier to EVs. Research using lipopolysaccharide (LPS) to activate innate immunity showed enhanced EV uptake across the BBB independently of structural disruption, suggesting that neuroinflammation modulates transcytosis rates through inflammatory signaling pathways. This has practical implications: diseases that compromise the BBB may produce a measurable spike in circulating brain-derived EVs, which could itself serve as a diagnostic signal.
Alternative Transit Routes
Beyond the classical BBB route, two additional anatomical pathways for brain-to-blood EV transit have been proposed. The meningeal lymphatic vessels, recently discovered to connect the CNS to the peripheral lymphatic system, may provide a conduit for EVs to enter peripheral circulation. Additionally, the blood–CSF barrier at the choroid plexusrepresents a third checkpoint through which brain-derived EVs may gain systemic access. These alternative routes remain underexplored but open exciting research avenues for enhancing EV biomarker yield from blood samples.
Key Biomarkers Carried by Brain-Derived EVs
The true diagnostic power of brain-derived EVs lies in their cargo. Because EV formation is a tightly regulated biological process reflecting the metabolic and molecular state of the parent cell, the molecules packaged within EVs are not random — they carry disease-specific information that mirrors concurrent changes happening within brain tissue.
Tau and Phosphorylated Tau
Tau is a microtubule-associated protein that, when hyperphosphorylated, forms the neurofibrillary tangles characteristic of Alzheimer’s disease. Multiple studies have demonstrated elevated levels of total tau, phosphorylated tau-181 (p-tau-181), and phosphorylated tau-231 (p-tau-231) in plasma neuron-derived EVs in AD patients compared to controls. Critically, these EV-associated tau species were found to negatively correlate with cognitive scores, providing a direct link between circulating EV cargo and clinical disease progression. Even in individuals with cognitive decline less severe than mild cognitive impairment, higher tau and p-tau levels have been detected in NDEVs compared to cognitively stable individuals.
Amyloid-Beta Peptides
Amyloid-beta (Aβ) peptides, particularly Aβ42 and the Aβ42/Aβ40 ratio, are established hallmarks of AD and have been detected in plasma EVs at levels correlated with those found in CSF. EVs appear to facilitate the intercellular spread of Aβ between neurons, meaning they serve both as biomarker carriers and as participants in disease propagation — a duality that makes their study particularly critical.
Alpha-Synuclein
In Parkinson’s disease, the protein alpha-synuclein (α-syn) misfolds and aggregates into Lewy bodies. Plasma exosomes containing CNS-derived α-syn have been successfully isolated using L1 cell adhesion molecule (L1CAM)-based immunoaffinity capture protocols, demonstrating that α-syn-carrying EVs are genuinely CNS-derived and elevated in Parkinson’s patients. L1CAM has emerged as a reliable surface marker for isolating neuron-specific EVs from blood, enabling enrichment of the brain-derived EV fraction from a complex systemic background.
mRNA and miRNA Cargo
Beyond proteins, EVs carry functional RNA molecules. A landmark 2024 study from Johns Hopkins University demonstrated that blood EVs carrying brain-specific mRNAs could be used to infer transcriptional changes occurring in the brain. By analyzing 13 female brain-specific transcripts in circulating EVs, researchers found associations with postpartum depression, establishing that EV mRNA levels can reflect gene expression states in the brain at a distance. Similarly, microRNAs (miRNAs) within brain-derived EVs serve as powerful epigenetic biomarkers, with specific miRNA profiles corresponding to neurological disease states and their targets showing pronounced expression in brain tissue.
Synaptic Proteins
Synaptic integrity is among the earliest casualties in neurodegenerative disease, predating neuronal death by years. Blood neuro-EV proteins including GAP43 (growth-associated protein 43), neurogranin, SNAP25, and synaptotagmin have been explored in combination panels as synaptic biomarkers. Declining levels of these synapse-specific proteins in plasma neuronal exosomes have been linked to progressive cognitive impairment and AD stage, suggesting that EV-based synaptic profiling may enable staging of neurodegenerative disease with greater sensitivity than existing methods.
APLP1 and Novel Surface Biomarkers
Amyloid precursor-like protein 1 (APLP1) represents a newer generation of brain-specific EV surface biomarkers. Because APLP1 is predominantly expressed in oligodendrocytes and neurons, EVs bearing APLP1 on their membrane can be selectively isolated from peripheral blood using APLP1-specific antibodies, enabling a clean separation of brain-derived EVs from the much larger pool of EVs shed by other organs. Validation using transgenic mouse models confirmed APLP1 coexpression with neuron-specific markers, underscoring its transformative potential for non-invasive brain disease diagnostics.
EV-Based Liquid Biopsy: Clinical Applications and Current Evidence
The concept of liquid biopsy — diagnosing disease through analysis of biological markers in easily accessible bodily fluids — is well established in oncology through circulating tumor DNA and circulating tumor cells. Brain-derived EVs extend this paradigm to neurological disease, where tissue biopsy is clinically impossible and CSF collection remains burdensome.
Alzheimer’s Disease
Brain-derived EV biomarkers have shown exceptional promise in Alzheimer’s research. Studies have demonstrated that the combination of p-tau-181, Aβ42 levels, and the tau/Aβ42 ratio in plasma neuron-derived EVs can distinguish between controls, mild cognitive impairment, and AD with accuracy approaching CSF-based measurements. This is significant because CSF collection requires a lumbar puncture, whereas EV analysis requires only a blood draw. Clinical trials are underway at multiple institutions to validate EV-based biomarker panels as screening tools for AD in at-risk populations.
Parkinson’s Disease
The ability to capture CNS-derived α-syn from peripheral blood via L1CAM-positive EV isolation has opened a new chapter in Parkinson’s diagnostics. Because current Parkinson’s diagnosis relies heavily on clinical observation of motor symptoms — which appear only after significant neurodegeneration has already occurred — blood EV biomarkers could enable detection years earlier, when neuroprotective interventions may be most effective.
Psychiatric Disorders
The Johns Hopkins group’s work on EV mRNAs as brain transcriptome proxies is particularly relevant for psychiatry, where objective molecular biomarkers are almost entirely lacking. The demonstration that EV mRNA levels from peripheral blood can infer transcriptional changes in the brain represents a paradigm shift for conditions such as depression, bipolar disorder, and schizophrenia, where brain biopsies are ethically and practically impossible.
Brain Tumors and Glioblastoma
Tumor-derived EVs are actively released into circulation from glioblastoma (GBM) and other brain cancers, either through passive BBB leakage caused by tumor invasion or through active transcytosis. A 2025 Nature Communications study from a Phase I clinical trial demonstrated that EVs released following controlled BBB opening in GBM patients — using low-intensity pulsed ultrasound with microbubbles — could serve as biomarkers of treatment response, enabling real-time monitoring of tumor cell death through blood-based EV analysis.
article: how to extract meaningful, brain-specific molecular signals from a peripheral blood sample that is dominated by vesicles from non-neuronal sources.
Technical Challenges and Standardization
Despite the compelling biology, the translation of brain-derived EV biomarkers from bench to clinic faces several significant hurdles that the field is actively working to address.
Isolation and enrichment: The brain contributes only a small fraction of total circulating EVs — the majority are shed by platelets, red blood cells, endothelial cells, and immune cells. As reviewed in PLoS Medicine and other journals, extracting the brain-specific EV fraction requires immunoaffinity enrichment using neural surface markers (L1CAM, NCAM, APLP1), which adds complexity and cost to the workflow.
Standardization: EV isolation methods range from ultracentrifugation and size-exclusion chromatography to polymer precipitation and acoustic trapping. Each method yields EVs of different purity and size distributions, making cross-study comparisons difficult. The Minimal Information for Studies of Extracellular Vesicles (MISEV2018) guidelines represent the community’s attempt to harmonize methodology, but implementation remains inconsistent.
Sensitivity of detection: Pathological brain proteins carried within EVs exist at very low concentrations in blood, often below the detection thresholds of conventional immunoassays. Advances in single-molecule detection platforms (such as Simoa and digital ELISA) have dramatically improved sensitivity, but widespread clinical deployment requires further cost reduction and workflow simplification.
CNS specificity of EV surface markers: L1CAM, long used as a neuron-specific EV marker, has come under scrutiny following evidence that it is also expressed by non-neuronal cells. This underscores the need for validated, rigorously brain-specific surface markers — APLP1 being a promising candidate — to ensure that isolated EVs genuinely reflect brain biology
The Future: Where EV Biomarker Research Is Heading
The EV biomarker field is evolving rapidly, driven by improvements in both molecular biology tools and clinical study design. Several emerging directions hold particular promise.
Multi-marker EV panels: Rather than relying on single biomarkers, researchers are developing panels combining protein, RNA, and lipid data from brain-derived EVs to achieve high diagnostic accuracy. Combining tau, p-tau-181, Aβ42, and synaptic proteins with miRNA signatures may yield sensitivity and specificity approaching that of PET imaging at a fraction of the cost.
EV-based monitoring of treatment response: As disease-modifying therapies for AD, Parkinson’s, and other neurodegenerative conditions enter clinical trials, there is an urgent need for biomarkers that can dynamically track treatment effects. Brain-derived EV biomarkers, which reflect real-time molecular changes in the brain, are ideally positioned for this role — enabling trial teams to verify target engagement weeks rather than years into a study.
Artificial intelligence and EV proteomics: Machine learning algorithms applied to large EV proteomics or transcriptomics datasets have the potential to identify novel brain-specific EV signatures that would be invisible to hypothesis-driven research. Integration of these approaches with large biobanks is accelerating biomarker discovery.
Engineered EVs as theranostic agents: Beyond diagnostics, brain-derived EVs are being engineered as drug delivery vehicles back into the CNS — exploiting the same BBB-crossing mechanisms that allow endogenous brain EVs to enter the blood. This creates the tantalizing possibility of theranostic EVs that simultaneously carry therapeutic cargo inward and diagnostic biomarkers outward.
Conclusion
Extracellular vesicles represent one of the most exciting frontiers in modern neuroscience and clinical diagnostics. By acting as molecular couriers — packaging brain-specific proteins, RNAs, and lipids and carrying them across the blood–brain barrier via active transcytotic mechanisms into the peripheral circulation — brain-derived EVs make the previously inaccessible brain readable through a simple blood test.
The evidence reviewed here demonstrates that EV-associated biomarkers can reflect the molecular pathology of Alzheimer’s disease, Parkinson’s disease, glioblastoma, traumatic brain injury, and psychiatric conditions with a specificity and sensitivity that challenge conventional approaches. While significant technical challenges around isolation standardization, detection sensitivity, and marker validation remain, the pace of progress in the field suggests that EV-based blood biomarkers for neurological disease will enter routine clinical practice within the coming decade.
For patients, clinicians, and researchers alike, the promise is clear: the brain, once a molecular black box, is learning to speak through the language of extracellular vesicles — and we are finally learning to listen.



Leave a Reply