
Bio Techne Ella: Accelerate Biomarker Analytics with bio techne ella
If you’ve ever spent days wrestling with a traditional ELISA, you know the frustration. The endless pipetting, the meticulous wash steps, the nagging worry about operator-to-operator variability—it’s a time-consuming process that can easily compromise your data. What if you could get those same results, but with far greater precision and in under 90 minutes?
That’s exactly what the Bio-Techne Ella platform delivers. It’s a completely hands-free, automated immunoassay system that takes the manual labor and guesswork out of protein quantification.
How the Bio-Techne Ella Platform Works
Think of a traditional, multi-step immunoassay. Now, imagine condensing all those steps—the reagent additions, incubations, washes, and final reads—into a self-contained, automated process. That’s the principle behind Ella.
The real innovation lies within its proprietary Simple Plex™ cartridge. This isn’t just a plate; it’s a miniaturized, single-use laboratory. Each cartridge contains all the necessary reagents and microfluidic channels needed to run a complete assay, pre-calibrated and ready to go.
The Power of Walk-Away Automation
The workflow is beautifully simple. You just add your sample to the cartridge, place it into the Ella instrument, and press start. From there, the machine handles everything.
- Precise Fluidics: The system automatically moves your sample through the microfluidic circuits, ensuring every reaction happens under perfectly controlled conditions.
- Automated Steps: All wash steps and incubation periods are timed and executed by the instrument, eliminating the human error that plagues manual methods.
- Built-in Reads: The final signal is detected internally, generating clean, quantifiable data without any user intervention.
- Instant Analysis: Ella automatically creates standard curves, calculates analyte concentrations, and spits out a full quality control report.
This end-to-end automation does more than just free up your time. It tackles one of the biggest challenges in biomarker research: the operator-to-operator variability that can undermine the integrity of long-term studies and multi-site clinical trials.
By standardizing the entire workflow into a “sample-in, data-out” process, the Bio-Techne Ella instrument produces results that are not only fast but also exceptionally consistent. It’s a critical advantage when data reproducibility is non-negotiable.
Designed for Speed and Simplicity
The entire platform is built for efficiency. A full run is typically finished in less than 90 minutes, a dramatic improvement over manual assays that can stretch across hours or even days.
For labs needing to scale up their analytical throughput without adding headcount, Ella offers a powerful solution. While it excels at streamlining protein quantification, the same principles of automation and precision are vital for protein characterization. You can see how this applies to other platforms in our deep dive on the ProteinSimple Jess.
Key Assay Capabilities for Neurological Biomarkers
To really grasp what makes the Bio-Techne Ella platform a game-changer for neurology, you have to look past the automation and dig into its core performance. It all comes down to three things: sensitivity, dynamic range, and multiplexing. These aren’t just specs on a data sheet; they’re the solution to the persistent headaches of working with tricky neurological biomarkers.
Exceptional Sensitivity: Detecting the Undetectable
The first major hurdle in neurology research is signal. Key biomarkers, especially those tucked away in neuron-derived exosomes (NDEs), often float around at minuscule concentrations—down in the picogram-per-milliliter range. Most standard assays are simply blind to these faint whispers, leaving you with frustratingly ambiguous or unquantifiable data.
Ella’s microfluidic design cuts right through this noise. Think of it as having a high-powered telescope that can resolve distant stars completely invisible to the naked eye. It’s engineered to consistently pick up on these ultra-low-abundance proteins, turning what was once a blurry mess into a clear, quantifiable result.
Wide Dynamic Range for Comprehensive Analysis
Next up is the platform’s impressive dynamic range. Traditional immunoassays often put you in a bind: you can either set them up to measure very low concentrations or very high ones, but rarely both in the same go. This forces you into a tedious cycle of sample dilutions, which eats up precious sample material, introduces pipetting errors, and wastes valuable time.
The Ella system sidesteps this problem entirely with a dynamic range stretching over 4 logs. This means you can accurately quantify both a low-level baseline protein and a highly expressed pathological marker from the exact same, undiluted sample.
This is a massive advantage in neurological studies, where biomarker levels can swing wildly between a healthy control and a patient with advanced disease. You capture the full biological picture in a single run, no compromises needed.
This ability is absolutely critical for tracking disease progression or measuring a drug’s effect, where you expect to see biomarker concentrations shift by orders of magnitude.
Maximizing Insights with Multiplexing
Finally, there’s Ella’s powerful multiplexing capability. Neurological diseases are almost never about a single rogue protein; they are complex symphonies of multiple, interconnected pathways. To truly understand what’s going on, you need to see how multiple players are behaving at the same time, in the same sample.
Ella lets you analyze up to eight different proteins simultaneously from a tiny amount of sample. This is a lifesaver when you’re working with irreplaceable samples like cerebrospinal fluid (CSF) or purified exosomes. Instead of burning through your material running eight separate ELISAs, you get a full panel of results from one efficient, hands-off run.
For instance, a researcher could measure Tau, pTau, Aβ40, and Aβ42 alongside key inflammatory markers like IL-6 and TNF-alpha, all from one plasma sample. This integrated dataset provides a far richer biological context than any single-analyte measurement ever could.
To put it all together, here’s a quick comparison that highlights just how different the workflow and data quality can be between Ella and traditional plate-based ELISAs.
Bio-Techne Ella vs. Traditional ELISA: A Performance Snapshot
This table contrasts the key performance characteristics of the Ella platform with conventional ELISA methods, demonstrating Ella’s advantages for demanding biomarker applications.
| Feature | Bio-Techne Ella | Traditional ELISA |
|---|---|---|
| Sensitivity | Sub-picogram/mL detection, ideal for low-abundance biomarkers. | Often limited to nanogram/mL range, misses faint signals. |
| Dynamic Range | >4 logs, measures low and high concentrations in one run. | 1-2 logs, requires multiple dilutions and repeat runs. |
| Multiplexing | Up to 8 analytes per sample, providing rich pathway data. | Typically single-analyte, consuming more sample and time. |
| Hands-On Time | <15 minutes per run, fully automated process. | 2-4 hours of manual pipetting, washing, and incubating. |
The differences aren’t subtle. For labs focused on the complex, low-abundance biomarkers typical of neurodegenerative disease, the technical advantages of a platform like Ella directly translate into more reliable, comprehensive, and ultimately more insightful data.
How Ella Redefines Your Research Workflow
Switching to the Bio-Techne Ella platform is more than just a simple upgrade from traditional immunoassays. It’s a fundamental shift in how your lab gets work done. Say goodbye to the days of dedicating a skilled technician to hours of meticulous, error-prone pipetting. With Ella, the entire process becomes a straightforward “sample-in, results-out” model that dramatically improves your lab’s efficiency.
Think about a typical day in a busy neurology lab. Your team has just finished processing a batch of precious patient samples—maybe it’s plasma, serum, or even purified neuron-derived exosomes from a platform like NeuroDex’s ExoSort. Instead of spending the rest of the day mixing buffer solutions, coating plates, and navigating complex dilution series, your workflow gets astonishingly simple. You just add the sample directly into a pre-configured cartridge and slide it into the instrument.
From Manual Labor to Walk-Away Automation
The moment you press “start,” your job is essentially done. The Ella instrument handles everything else on its own, managing every wash, incubation, and reagent step with machine precision. This level of automation delivers two massive advantages for any research team.
- Drastically Reduces Hands-On Time: The entire process, from loading your sample to getting fully analyzed data, requires less than 15 minutes of active user involvement.
- Virtually Eliminates Human Error: By removing all the manual steps, Ella minimizes the run-to-run variability that can compromise data quality, ensuring your results are consistent and reliable.
This walk-away capability frees up your highly trained staff to focus on higher-value work, like designing experiments, interpreting data, and planning the next critical phase of your research program. Your team can drive the science forward instead of being tethered to the bench.
Data Analysis That Works for You
The efficiency gains don’t end when the assay run is finished. The Bio-Techne Ella platform automatically generates a complete data report, with standard curves, calculated analyte concentrations, and detailed QC metrics all included. This presentation-ready output saves countless hours that would otherwise be lost to manually plotting curves and crunching numbers in separate software.
The platform delivers fully analyzed results, empowering researchers to make decisions faster. It transforms the assay process from a multi-day chore into a streamlined, automated workflow that produces reliable data in under 90 minutes.
This combination of speed, simplicity, and data quality is what makes the platform so powerful. The infographic below visualizes the core capabilities—sensitivity, dynamic range, and multiplexing—that make this workflow possible.
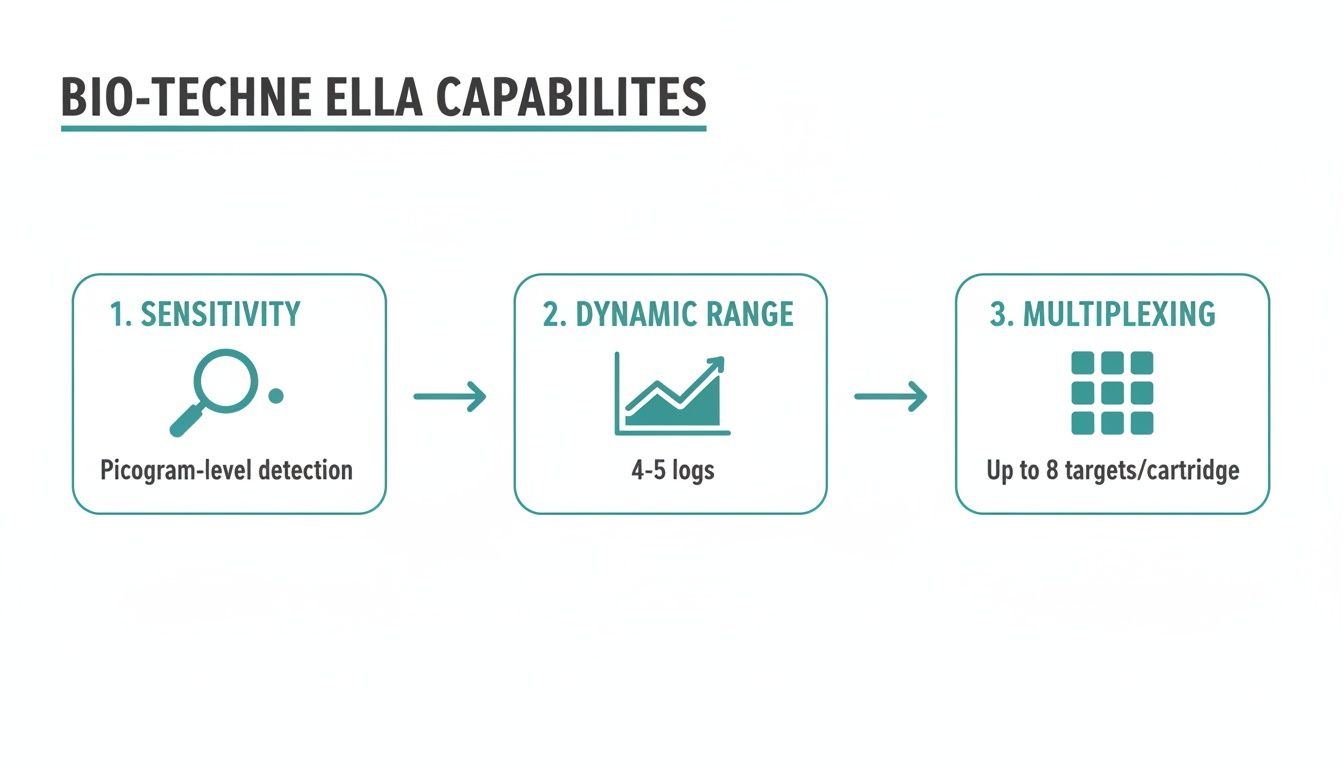
These features are essential for meeting the growing demand for reliable neurological diagnostics. The global neurological biomarkers market, valued at USD 9.64 billion in 2022, is projected to climb past $10.5 billion by 2028, fueled by the urgent need for better tools in diseases like Alzheimer’s and Parkinson’s. You can learn more about these drivers in the full neurological biomarkers market report. By optimizing critical lab workflows, platforms like Ella are enabling researchers to meet this challenge head-on.
Accelerating Clinical Trials with Reliable Biomarker Data
Taking a promising compound from the lab bench into a clinical trial is a monumental leap, and the quality of your data is everything. This transition is exactly where the Bio‑Techne Ella platform proves its worth, bridging the gap between exploratory research and the high-stakes world of pharmaceutical development. Its precision and reproducibility aren’t just “nice-to-haves”—they’re what it takes to generate the kind of robust biomarker data that regulatory bodies demand.
Ella’s role is felt across every phase of a clinical trial. In the early stages, it’s a game-changer for patient stratification. By accurately measuring key biomarkers like NF-L or pTau directly from blood, sponsors can be sure they’re selecting the right patient cohorts, dramatically increasing the odds of demonstrating a therapeutic effect.
Standardizing Data Across Multi-Site Studies
One of the biggest headaches in large-scale clinical trials is keeping data consistent across different testing sites. When manual assays are performed by different teams in different labs, that inter-lab variability can create enough noise to completely obscure a real treatment signal. The standardized, automated workflow of the Bio‑Techne Ella platform was designed to solve this exact problem.
Because the entire immunoassay is self-contained within a pre-calibrated, single-use cartridge, the results coming out of a lab in Boston are directly comparable to those from a lab in Berlin. This machine-driven consistency gives clinical sponsors immense confidence in their data and helps de-risk the entire development pipeline. As you navigate these projects, it’s also helpful to see the bigger picture, including the critical role of Medical Affairs in pharma in communicating this kind of scientific value.
By eliminating operator-dependent variability, Ella ensures that any observed changes in biomarker levels reflect true biological responses to a therapeutic, not differences in how the assay was run. This is a crucial advantage for making confident go/no-go decisions.
This standardized approach isn’t just an improvement; it’s a strategic imperative. By 2026, pharma and biotech companies are expected to hold nearly 50% of the neurological biomarkers market, using these tools to sharpen their drug discovery efforts. Biomarker-stratified trials can shrink enrollment needs by 20-30% and significantly boost success rates, helping bring the historic 90% failure rate in neurodegenerative studies down to under 70%.
Powering Pharmacodynamic and Endpoint Analysis
Beyond just selecting patients, Ella is an incredibly powerful tool for measuring pharmacodynamic (PD) responses. Its high sensitivity and wide dynamic range give research teams the ability to track how a drug engages its target and modulates downstream biological pathways over time.
This capability is absolutely central to demonstrating a drug’s mechanism of action and finding the optimal dose. For companies looking to assemble a rock-solid data package for regulatory submission, the reliable, reproducible data generated by Bio‑Techne Ella is indispensable.
At NeuroDex, we specialize in integrating these advanced analytics into our comprehensive clinical trial services to support every phase of your program.
Navigating Assay Validation and Regulatory Compliance
Getting a groundbreaking biomarker out of the research lab and into a clinical setting is a huge leap. It’s a journey that demands meticulous assay validation and strict adherence to quality systems—a path that can feel overwhelmingly complex and slow. This is precisely where the automated, standardized design of the Bio‑Techne Ella platform comes in, helping to smooth out the bumps on the road to regulatory approval.
The system’s built-in consistency is a massive advantage when operating under Good Laboratory Practice (GLP) principles. Think about it: every single step of the immunoassay happens inside a self-contained cartridge, all automated. This design practically eliminates the operator-to-operator variability that can plague manual assays, creating an incredibly stable and reproducible testing environment. That rock-solid foundation is non-negotiable for any successful validation study.
Meeting GLP and CLIA Standards
For an assay’s data to be trusted in clinical decisions, it must be accurate, reliable, and completely traceable. The Bio‑Techne Ella system is built to deliver on this, generating detailed, automated reports for every single run. These reports capture everything—standard curves, quality control data, and final concentrations—all without manual transcription. This built-in documentation is a lifesaver, streamlining the creation of the robust validation package required for regulatory submissions to bodies like the FDA or EMA.
Its performance also makes it a workhorse in CLIA-certified diagnostic labs. Since CLIA (Clinical Laboratory Improvement Amendments) regulations govern all human laboratory testing in the U.S., consistency and reliability are paramount. Ella’s ability to produce high-quality results with minimal hands-on time is a perfect fit for the high-throughput, quality-driven world of a CLIA lab.
By providing a robust, automated foundation, the Ella platform helps de-risk the regulatory pathway for new biomarkers. This gives clinical trial sponsors the confidence they need to adopt next-generation diagnostics for their programs.
You can’t overstate how critical this is, especially in neurology. Neurological biomarkers are on track to make up over 25% of the entire diagnostics market by 2026, with pharma companies using them to cut trial times by as much as a quarter. With over 80% of neuro trials expected to require biomarkers by 2030, CLIA-validated services are the essential bridge between promising research and actual patient care. At NeuroDex, our deep expertise in these standards is a cornerstone of our biotech vendor services.
Finally, a modern lab’s compliance strategy has to look beyond just the assay itself and consider its larger operational footprint. Incorporating sustainable laboratory practices is quickly becoming a core part of being a responsible, forward-thinking organization.
Partnering with NeuroDex to Unlock Your Biomarker Strategy

Knowing what a powerful instrument like the Bio‑Techne Ella can do is one thing. The real work—and where most programs face hurdles—is weaving that power into a complete, end-to-end biomarker strategy that delivers clear, actionable results for your drug development program. This is precisely where a partnership with NeuroDex makes the difference.
We don’t just run assays. We integrate the raw analytical muscle of the Bio‑Techne Ella with our own specialized technologies, creating a solution purpose-built for the unique demands of precision neurology.
By itself, Ella gives you incredible speed, sensitivity, and reproducibility. But when we pair it with our proprietary ExoSort technology for isolating neuron-derived exosomes (NDEs), it becomes an even more focused tool for interrogating brain biology from a simple blood sample. This synergy is the foundation of our entire approach.
Your End-to-End Solution Partner
We’re not a fee-for-service lab that just sends back data. We provide an integrated pathway from initial discovery all the way to clinical implementation. Our expertise covers every critical stage needed to move your program forward:
- Discovery: Identifying novel neurological biomarkers from the precious cargo inside NDEs.
- Validation: Rigorously testing assay performance to meet demanding GLP standards.
- Implementation: Deploying those validated assays in CLIA-certified environments ready for clinical trial use.
This holistic model is designed to de-risk your development pipeline. We handle the complexities of assay validation and regulatory compliance so your team can stay focused on the science and the results.
A partnership with NeuroDex provides more than just access to the Bio‑Techne Ella platform. It gives you a dedicated team of experts committed to translating complex neurological biomarkers into clear, reliable data that accelerates therapeutic development.
For pharmaceutical companies, biotechs, and CROs, this means making faster, more confident decisions. When you work with us, you gain a partner that deeply understands the science, the technology, and the regulatory landscape required to power the future of precision neurology.
Your Questions Answered
We get a lot of questions about how the Bio‑Techne Ella platform works in a real-world lab setting. Here, we tackle some of the most common ones to give you a clear picture of how it can fit into your biomarker analysis pipeline.
Can Ella Handle Tricky Samples Like Neuron-Derived Exosomes?
Yes, absolutely. This is one of the areas where the Bio‑Techne Ella really shines. It’s built to handle the complexities of biological samples like serum, plasma, CSF, and the purified exosome preps we specialize in.
The platform’s microfluidic design and incredible sensitivity are perfectly matched for measuring the low-abundance proteins we often find in neuron-derived exosomes (NDEs). These are exactly the kinds of targets that can be a real headache for traditional assays. This makes Ella the ideal analytical engine to pair with advanced isolation methods like NeuroDex’s ExoSort platform, letting us dig deep into neurological pathways from nothing more than a simple blood draw.
How Does Bio-Techne Ella Stack Up Against Other Immunoassay Platforms?
Compared to a traditional ELISA, the difference is night and day. A manual ELISA can tie up a technician for over four hours of hands-on work. With Ella, you’re looking at less than 15 minutes of setup time to get fully analyzed results in under 90 minutes. It’s a massive leap forward in speed, precision, and efficiency.
When you put it next to other multiplex platforms, Ella’s main advantage is its sheer simplicity and reproducibility. The all-in-one cartridge design gets rid of the complicated troubleshooting and optimization that often comes with bead-based or planar array systems.
Ella’s key strength is its “sample-in, result-out” automation. This all but eliminates the operator-to-operator variability that can torpedo data integrity, especially in multi-site clinical trials or long-term studies.
What’s the Advantage of Working with an Integrated Service Provider?
Partnering with an integrated provider like NeuroDex means you’re getting a complete solution, not just a spreadsheet of raw data. Instead of just running samples and handing them back, we guide you through the entire process.
This starts with expert advice on designing your experiment, moves to specialized NDE isolation, and finishes with running your samples on the Bio‑Techne Ella under strict GLP/CLIA quality standards. This soup-to-nuts approach ensures your biomarker data is not only accurate but also meaningful and ready for regulatory scrutiny, significantly shortening your journey from discovery to the clinic.
Ready to accelerate your neurological biomarker research with a truly integrated, end-to-end solution? Partner with NeuroDex Inc. to combine specialized exosome isolation with the power of the Bio-Techne Ella platform.

Leave a Reply